7 Apr 2026
Jacqui Matthews BVMS, PhD, FRSE, FRCVS explains how the use of tools such as risk assessments can help to reduce helminth infections while also reducing anthelmintic use.

Image: Mark J. Barrett / Adobe Stock
Equine gastrointestinal helminths are widespread, with cyathostomins (small strongyles and small redworms) and the tapeworm, Anoplocephala perfoliata, being the most prevalent.
In the UK, large strongyle infections have become rarer, largely due to the widespread use of broad-spectrum anthelmintics.
Other parasites to consider are Parascaris species, which primarily affect foals, and pinworms (Oxyuris equi). The overuse of anthelmintics has led to high resistance rates in cyathostomins and Parascaris species, while treatment efficacy against O equi has diminished1.
Reports are also emerging of treatment failures of anti-tapeworm products2. The increase in reports demonstrating reduced anthelmintic effectiveness underscores how difficult it is to control equine helminth infections successfully, yet prevent resistance from developing at the same time.
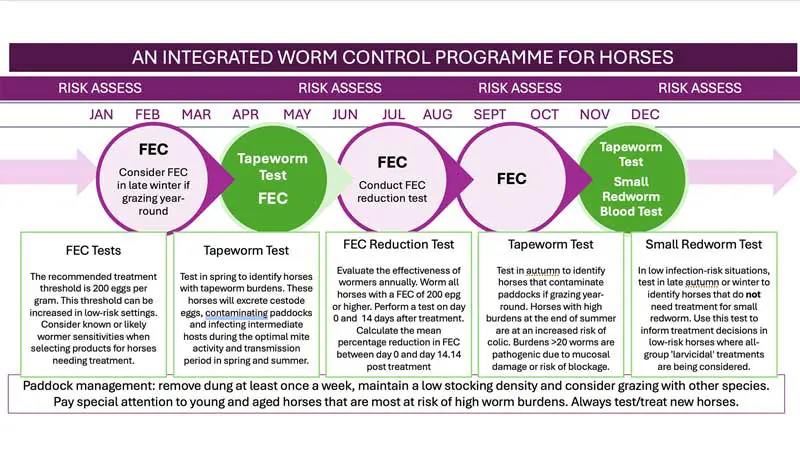
Modern strategies for managing these parasites must, therefore, prioritise reducing treatment frequency by combining integrated pasture management with diagnostic testing, rather than relying on routine, blanket worming. Conducting a thorough risk assessment is the critical first step in this approach.
A risk-based strategy for helminth control (Figure 1) requires thorough evaluation of the individual, its management and, if available, recent test results. Key considerations include the following.

Younger horses exhibit greater susceptibility to helminths, tend to harbour larger burdens and are more at risk of disease.
Foals are at risk of harbouring significant ascarid infections – especially up to eight months of age. Horses aged up to four years are more susceptible to cyathostomins and will excrete more worm eggs3, increasing the infection risk for any co-grazing older companions. Additionally, older horses – especially those affected by pituitary pars intermedia dysfunction – may experience increased susceptibility to helminths.
A history of worm-associated disease – especially in the previous 24 months – is an important risk factor.
Key clinical signs that may indicate a risk of helminth infection include colic episodes, diarrhoea or weight loss – especially when supported by direct evidence of parasites being present.
Horses that graze for prolonged periods, or year round, face an elevated risk of parasitic infection.
Comprehensive risk assessments should account for both the duration of grazing and seasonal variables. Parasite exposure varies seasonally, with peak transmission observed from late spring through early autumn in the UK, coinciding with environmental conditions that favour optimal development of pre-parasitic stages.
Effective paddock management reduces environmental contamination and infection risk:
Dung removal: regular removal of faeces from paddocks reduces infection risk by significantly lowering strongyle transmission4. Research indicates that removing dung twice weekly can decrease strongyle larvae levels by more than 90 per cent compared to untreated paddocks, while also increasing usable grazing area by approximately 50 per cent5.
Keep in mind that placing dung heaps close to paddocks increases the risk of transmission, as larvae and tapeworm-infected oribatid mites can migrate from the dung heap back on to fields.
Stocking density: high densities of horses increase contamination and, therefore, increase the level of helminth transmission and risk on paddocks.
Paddock resting: permanent equine paddocks – especially those permanently containing young horses – carry the highest risk of helminth transmission.
Grazing with cattle or sheep: alternate grazing with non-equine species can reduce strongyle transmission6, although ruminants may carry liver fluke (Fasciola hepatica), a risk that needs to be assessed.
Harrowing: under typical UK conditions, harrowing generally disperses worms across paddocks rather than eliminating by promoting desiccation. Therefore, harrowing does not reduce infection risk.
New horses or those returning from grazing elsewhere can introduce parasites, including resistant ones. Frequent introductions, time spent outside normal fields and a lack of quarantine procedures all increase risk.
Consistent test and treat, or treat only, quarantine protocols will help lower the chance of introducing new or resistant worms.
Test results are key to assessing risk by indicating contamination levels and exposure to infection. A higher percentage of horses testing positive generally means a greater risk of helminth infection in the population.
Faecal egg count (FEC) results: in herds that are well managed, less than 20 per cent of horses are responsible for shedding most of the eggs, making targeted treatments an effective strategy. Conversely, poorly managed herds tend to have more horses with high FECs, which increases both the risk of parasite transmission and the need for additional treatments. FEC testing is crucial for evaluating how well drugs work. If a FEC reduction test shows decreased effectiveness of anthelmintics, this represents a major risk factor.
Tapeworm antibody test results: these identify tapeworm burdens not detected by FECs7. In well-managed groups, the prevalence of tapeworm infection among horses is generally low or absent, whereas horses grazing poorly managed paddocks face a higher risk and are more likely to test positive for infection.
Small redworm antibody test results: this test detects infection by all cyathostomin stages8, including encysted larvae and male worms that FECs miss. In well-managed groups, most horses have low small redworm antibody levels (known as “serum scores”). However, horses grazing poorly managed paddocks – especially those younger than five years old – are more likely to have results above the 10,000-cyathostomin burden threshold.
Risk assessments are recommended at intervals of three to 12 months, determined by the risk category and changes in management or test outcomes. The online resource, What’s Your Worm Risk? (whatsyourwormrisk.com), offers structured guidance for assessing helminth infection risk using horse-specific answers to nine key questions.
In the UK, environmental conditions from mid-spring to late summer are the most conducive to parasitic helminth transmission.
Higher temperatures, humidity and rainfall all enhance larval development9. Therefore, pasture management practices should be optimal during these periods.
Effective grazing management practices involve maintaining stocking densities at more than one acre per horse, implementing pasture rotation or rest periods, and avoiding continuous grazing of horses on the same paddocks – particularly for younger groups.
Research has shown that when stocking density is higher, strongyle egg shedding also increases. Therefore, providing adequate paddock space for each group is crucial10.
Extending rest periods for paddocks – ideally a minimum of six months, especially from the start of the year – can lower strongyle infection risk; however, complete eradication of all worm stages may require up to two years11.
The environmental persistence of ascarid eggs and cysticercoid-infected mites is not yet fully understood. Grazing horses subsequent to ruminants has been demonstrated to reduce strongyle transmission6. Although Trichostrongylus axei can be transmitted to horses from pastures previously grazed by ruminants, this stomach worm seldom causes disease in equines.
However, including ruminants in pasture management plans requires careful risk evaluation – especially due to the possibility of F hepatica infections. It is advisable to assess risks and, if necessary, conduct fluke testing before introducing ruminants to horse paddocks.
Harrowing tends to be ineffective in the UK, since strongyle larvae and other parasitic free-living stages are resilient to brief dry spells and can migrate from sheltered sites within faecal pats after rainfall.
FECs are an essential method for achieving sustainable control of equine helminths.
In well-managed horse groups, most eggs are shed by only a few horses3,12. This enables targeted treatment of those high-shedding individuals and helps reduce overall use of anthelmintics.
Testing frequency should be based on risk factors such as age, management and season, but generally needs to be done every 8 to 14 weeks. Standard treatment thresholds generally utilise 200 eggs per gram (epg), although higher thresholds may be appropriate in populations classified as low risk.
FECs do not correlate with total worm burdens – particularly when larval stages are predominant; consequently, it is advisable to employ additional diagnostic methods. Annual FEC reduction tests are recommended to evaluate the effectiveness of anthelmintic treatments.
Coprological techniques exhibit limited sensitivity for diagnosing tapeworm infections, primarily due to sporadic egg shedding from adult tapeworms and the common occurrence of immature or sterile adult parasites in individual infections13.
Antibody-based assays using blood and saliva samples offer more reliable identification of horses that require treatment. These tests categorise horses as “low”, “borderline” or “moderate/high”, with anti-tapeworm treatment advised for individuals falling within the latter two results categories.
Biannual testing is recommended for horses at increased risk of infection; for example, horses between one and five years of age or those managed in environments with suboptimal paddock practices and/or a history of tapeworm-related disease.
Conducting tests in spring facilitates targeted treatment of infected horses, thereby reducing egg dissemination during periods when oribatid mites – the intermediate hosts – are most active on pasture14.
Autumn testing assists in identifying horses that have acquired tapeworm burdens during the peak summer transmission season, allowing for timely treatment to mitigate the risk of colic.
In groups considered low risk, the small redworm blood test8 helps identify horses that do not require larvicidal therapy for cyathostomin infection in autumn or winter.
Eligible horses should be classified as low risk for helminth infection, with recent FEC results less than 200epg.
When applied according to these guidelines, the test facilitates significant reductions in anthelmintic use by identifying horses for whom treatment is unnecessary15.
This targeted approach reduces indiscriminate administration of anthelmintics to low-risk individuals, promoting sustainable control practices and reducing the risk of resistance development.
A reduction in anthelmintic treatments can lead to a rise in the prevalence of the large strongyle, Strongylus vulgaris16.
In situations where such treatments have been significantly reduced, it is advisable to use faecal culture tests to check for S vulgaris larvae. Westgate Labs now offers this testing service.
Special attention should be paid to this parasite in groups where macrocyclic lactone treatments have not been used for long periods.
Risk assessment, environmental management and targeted diagnostic testing form the essential foundations of effective equine parasite control programmes. Together, these measures (Figure 2) reduce reliance on routine anthelmintic treatments, help preserve the long-term efficacy of available drugs and lower pasture contamination by interrupting parasite life cycles. By reducing infection pressure and the associated risk of parasite-related disease, this integrated approach supports evidence-based, site-specific decision-making and promotes more sustainable equine helminth control across a wide range of management systems.
Jacqui Matthews qualified as a veterinary surgeon, completed a PhD in parasitology and then worked in academia for more than 25 years, leading a range of interdisciplinary projects focused on helminth infections of ruminants and horses. During this time, she taught many undergraduates and postgraduates in the subject area, and was awarded more than £13 million in competitive funding for research, from which more than 140 peer-reviewed papers have been published. Jacqui has written numerous lay articles, given seminars and workshops to stakeholders promoting sustainable helminth control practices, and for many years was technical advisor to the Control of Worms Sustainably in Cattle initiative and parasitology expert on the UK Veterinary Products Committee. She sits on several sub-groups of the CANTER initiative, set up to develop and promote best practice guidelines for parasite control in horses. One of her inventions – an ELISA test for equine small redworm – was commercialised by Austin Davis Biologics in 2019 and, in 2022, she moved to this company as director of veterinary science, where she leads the research and development programme.