14 Jul 2022
A bacterial equid disease predominately of the upper respiratory tract, strangles biosecurity and identifying infectious horses are key to preventing it. Development of a novel vaccine has added to the equine vet’s toolbox – as Philip Ivens explains.

Abscessation of local draining lymph nodes, and occasionally of the head, are strangles factors.
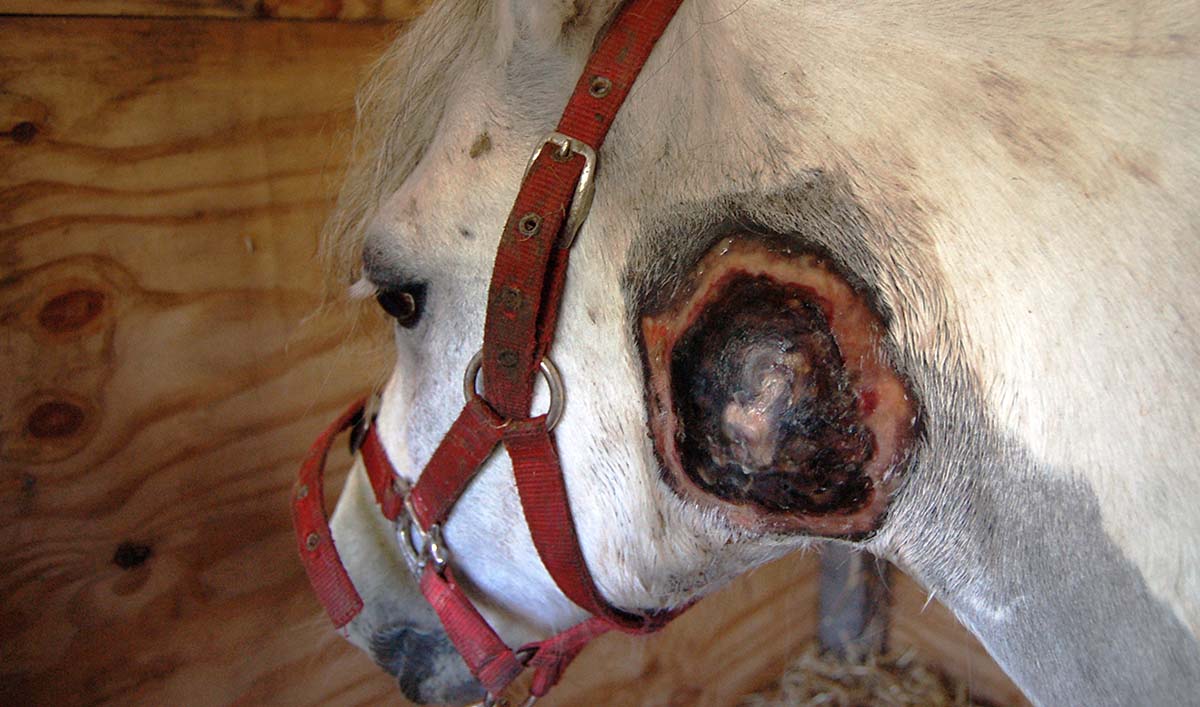
Strangles is a bacterial, predominately upper respiratory tract, disease of equids that causes abscessation of local draining lymph nodes and can, on occasion, cause external head abscessation.
The causative agent is Streptococcus equi subspecies equi, a member of the Lancefield group C, beta-haemolytic streptococci (Boyle et al, 2018). Biosecurity and identifying infectious individuals are key to preventing the disease. The exciting development of a novel vaccine to add to our armoury to combat this important equid disease will be discussed in detail in this article.
Working together, both as veterinary surgeons and as an equine industry as a whole, is vital to decrease the incidence of strangles, and to achieve the hypothetical – but scientifically possible – eradication of strangles.
The obligate equine pathogen modus operandi for long-term survival is developing a carrier state in recovered, clinically normal animals. Approximately 10% of animals that have had exposure to strangles will go on to develop a carrier state. The guttural pouch is the predominate place for carriage, although the sinuses are a theoretical possible (Newton et al, 1999).
The key to controlling strangles at the moment is identifying new acute cases, stopping movement of those horses, and isolating those affected and their in-contacts with rigorous biosecurity measures. Then, as the acute disease resolves, identifying horses that go on and become silent carriers, and treating those horses accordingly to prevent them posing an infection risk to naive horses in the future (Rendle et al, 2021). This is potentially costly and not achievable in some situations, due to economic and client reluctance. Therefore, potentially lowering the incidence of S equi infection through vaccination is desirable in this author’s opinion, in conjunction with existing control measures.
A full list of clinical signs of strangles is in Table 1. Rarer clinical signs include upper airway obstruction, and potential asphyxiation and death. More chronic sequelae include metastatic (bastard) strangles, purpura haemorrhagica and myositis.
The details of these sequelae are beyond the remit of this article, and details can be found in further reading (Boyle et al, 2018).
| Table 1. Clinical signs of strangles | |
|---|---|
| Classic strangles | Atypical strangles |
| Fever (>38.5°C) accompanied with depression and inappetence. | Mild short-term fever – may lose appetite for brief period of time. |
| Profuse yellow nasal discharge from both nostrils (thick mucus). | Mild serous/clear nasal discharge. |
| Swellings on the side of the head and nasopharynx which may burst and discharge pus. Can be hot and painful. | No abscess formation. |
| Sore throat – difficulty eating and extending head. | These horses are a risk to other horses and should be treated as infectious. |
| Dysphagia – from pressure/damage of the cranial nerves running through the floor of the GP. Can present like commonly seen oesophageal obstruction also known as “choke”. | Note: These clinical signs can be seen with a variety of respiratory diseases, not just strangles. |
The holy grail of S equi vaccination has been attempted a number of times before, although it has always been challenging and an ideal solution – in the author’s opinion – is yet to be reached. This article is going to focus on products licensed in the UK.
A live attenuated submucosal vaccine (Equilis Strep E; MSD Animal Health) has been registered for use in the UK since 2004.
The vaccine has single gene deletion aroA46-978bp of the TW 928 strain of S equi. IM vaccination provided 100% (3/3) protection from subsequent S equi challenge. However, severe injection site reactions precluded the use of this route for further studies.
A lip mucosa vaccination route was found to be safe in the study and acceptable levels of immunity were seen (Jacobs et al, 2000).
In the late part of the 20th century and early part of the 21st century, work in exploring a multi-component subunit vaccine expressing multiple surface proteins important to the virulence of S equi was undertaken (Flock et al, 2004 Flock et al, 2006). The work began in an era when a whole genome sequence of S equi was not known, and knowledge of the virulence factors for S equi was rudimentary.
Carefully, over time and with dead ends and cul-de-sacs along the way, brick by brick, eight proteins were identified, and three fusion proteins generated and incorporated into Strangvac, with a matrix V adjuvant, to give a broad and effective immune response to all known strains of S equi (Frosth et al, 2022; Robinson et al 2018; Robinson et al, 2020).
The author had the privilege to witness some of the 14 studies run at the, sadly, defunct Animal Health Trust across 14 years, while doing parallel research into S equi molecular epidemiology (Ivens et al, 2011).
Strangvac, the aforementioned vaccine, has no live agent, and has a novel technology of recombinant fusion proteins with a matrixV adjuvant. It is licensed for IM injection. The vaccine has DIVA capability, and will be culture and PCR-negative and seronegative on diagnostic strangles iELISAs.
It is for active immunisation of horses from five months of age in the UK (eight months in Europe) for the reduction of clinical signs in the acute stage of infection with S equi and reduction in the number of abscesses in the submandibular and retropharyngeal lymph nodes.
The first vaccination is from five months, and is 2ml by IM injection. The second vaccination is four weeks later.
A transient increase in body temperature of up to 2.6°C may very commonly occur and last for one to five days following vaccination, which will be seen by the owner as loss of appetite and demeanour changes in the first day post-vaccination.
Transient local tissue reactions are very commonly seen at the injection site, characterised by heat, pain and swelling (VMD, 2021) consistent with what is seen with other equine vaccines used in the UK, such as equine influenza vaccines.
A third vaccination three months after the second increases protection significantly (Robinson et al, 2020).
In horses at high risk of S equi infections, it is recommended to repeat the primary vaccination course after two months.
Data for prolonged clinical protection from the administration of single-dose revaccination is currently not available.
Based on measured antibody titres, immunological memory response was found in horses following repeated vaccination six months after primary vaccination (Robinson et al, 2020). The role of the measured antibodies in the immune response relevant for the protection against strangles is not known.
Undoubtedly, quick identification and diagnosis of new strangles cases, and isolating and ensuring the highest standards of biosecurity, will continue to be central to strangles control (Ivens and Pirie, 2021). Another vaccine entering the market with a more familiar route of administration and DIVA capability will strengthen our armoury in our continuous battle against S equi.
This author is sure our use of this vaccine will evolve and optimise over time as we get field data on its use in different clinical contexts. The scientific journey this vaccine has been on does not stop here, and it is incumbent on front-line clinicians to objectively assess the use of the vaccine to aid the iterative and/or evidence-based process to maximise the positive effect this vaccine will have in our fight to improve the welfare of horses from this disease.