24 Mar 2026
Al Manning BVetMed, MVetMed, GCLT, DipECBHM, MRCVS discusses the importance of everyone working together to improve environmental management in dairy herds.

Figure 1. An example of a cow with poor cleanliness score. This cow shows visible contamination over the feet, legs, udder and rump.
Data presented at the British Mastitis Conference has shown that the predominant mastitis epidemiology on UK dairy farms is “environmental”.
Of the farms signed up to receive QuarterPRO Mastitis Pattern Analysis Reports in 2024, 55% had an environmental lactation pattern (EL), 30% environmental dry period (EDP) 14% mixed and less than 1% contagious (Manning et al, 2024).
Cow cleanliness is an important driver of environmental infections, as many of the major pathogens causing mastitis are present in cow faeces. Once present on the teat surface, these opportunistic bacteria gain access to the streak canal when it is open, and can cause clinical or subclinical mastitis (Klaas and Zadoks, 2017). In EL pattern herds, the main risk period is around milking – immediately before, and for 30 minutes after. In EDP pattern herds, the biggest risk periods are in the first two weeks, and final two weeks of the dry period (Bradley and Green, 2004).
Herds with poor udder hygiene scores tend to see higher rates of mastitis (Barkema et al, 1998; Schreiner and Ruegg, 2003; Ellis et al, 2007; Dohmen et al, 2010). As a result, cow cleanliness scoring can be an important first step for a mastitis investigation. This article discusses assessment of cow cleanliness, and gives advice around improving cleanliness and minimising new infections.
Cow cleanliness can be subjective – most advisors will recognise where there is a problem with poor cow cleanliness on a unit, though farmers may become normalised to conditions on their farm. Several objective grading systems have been developed (Hughes, 2001; Schreiner and Ruegg, 2002; Reneau et al, 2005; Cook and Reinemann, 2007), these scoring systems are largely similar, and classify cows according to the area of faecal contamination over the udder, legs and flank.
Figure 1 shows an example cow with a poor cleanliness score – with visible dirt on the legs, udder and flank. Objective scoring systems can be useful for demonstrating that a farm has a problem with cow cleanliness, and monitoring improvement in response to changes in management. Further investigation of the environment is recommended where more than 10% of cows are visibly dirty.
The easiest place to score cow cleanliness is in the milking parlour, though observers may also wish to view cows in the collecting yard and post-milking yard. Cleanliness scoring is rarely a stand-alone visit: at the same time, an observer can also assess body condition, milk leaking, teat/udder lesions and the milking routine. Some auditors may also want to assess non-mastitis factors, for example, mobility scores, foot lesions or tail clipping. It is important to assess cow cleanliness at different times of the year and at different stages of the production cycle.
Even in herds that house all-year round, the environment is likely to change over the course of the year as temperature, humidity, rainfall, ventilation and management will all vary. In herds with a dry period mastitis epidemiology, cleanliness scoring of far-off and close-up dry cows is likely to be more useful.
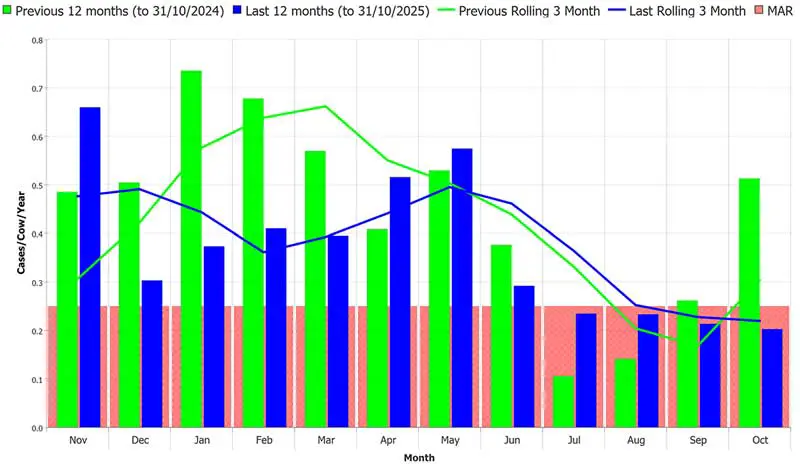
Where possible, cow cleanliness scoring should be targeted at the most “at-risk” periods according to analysis of clinical mastitis and somatic cell count data (Panel 1). Figure 2 shows mastitis incidence for a herd with a seasonal mastitis pattern – in this herd, cow cleanliness scoring should be carried out in the winter, as this is the main risk period for clinical mastitis. There is likely to be less benefit in cleanliness scoring in the summer as the mastitis rate is better controlled.

Bactoscan can also be a useful indicator of cow cleanliness. In herds with consistent poor cow cleanliness, the bactoscan is typically elevated above a target of 30 times 103 impulses/mL. Bulk milk bacteriology is likely to reveal increases across all bacteria types, particularly coliforms and psychrotrophs (Blowey et al, 1999). In the UK, average bactoscan is higher in the winter (Agriculture and Horticulture Development Board [AHDB, 2026a]), and this is likely caused by poorer cow cleanliness in housed herds.
If cows are coming into the parlour dirty, then in the short-term, it’s important that the parlour routine addresses this to minimise the risk of new infection and high bactoscan. The key stages of the pre-milking routine are commonly summarised as: strip, dip, wipe and attach. Any cows that are visibly dirty on entry to the milking parlour should have an additional cleaning step. Dirty teats should be washed with clean water – potable water should be free from faecal coliforms and Streptococcus (The Water Supply [Water Quality] Regulations 2016). It’s also important that the milking staff are clean – any hands that become contaminated with faecal material should be washed, and wearing gloves is likely to reduce contamination on milker’s hands.
Once teats are visibly clean, milkers should strip them to identify clinical mastitis, followed by application of a pre-milking teat disinfectant (MTD) to kill environmental bacteria on the surface of teats. Pre-MTD has been shown to reduce the number of environmental pathogens on teats (Munoz et al, 2008), and in resulting milk (Bradley et al, 2018). Ideally, teat dips should have a VM number, showing that they have proven efficacy. Pre-milking teat disinfection has been shown to be less effective for dirty teats (Munoz et al, 2008), so the aforementioned washing step remains important.
Following application of pre-MTD, the chemical should be wiped off using a clean paper towel or laundered cloth. This is an important step to dry the teats and to avoid chemical residues in milk (Breen, 2019). Clusters should be attached within 60 to 90 seconds of the start of teat stimulation, this reduces the risk of leaking milk that can contaminate the milking environment, and increases the risk of environmental mastitis (Schukken et al, 1990).
During the whole milking process, staff should minimise stress as this can lead to urination and defecation that will impair cow cleanliness further. Any clusters that become contaminated during milking should be washed with warm, potable water (as defined earlier in the article).
In the longer term, vets and advisors should work with their clients to improve cow cleanliness in lactating and dry cow environments. Readers are directed towards resources on environmental mastitis produced for the QuarterPRO initiative (AHDB, 2026b and 2026c). These articles address important areas of the lactating and dry cow environment that can relate to cow cleanliness. Panel 2 shows some key areas that relate to cow cleanliness and mastitis risk.

It is important that changes to the environment are targeted at the time where cow cleanliness is worst (and mastitis rates highest). Poor cow cleanliness can be influenced by problems in various areas of the farm.
Where data analysis points towards a particular problem with dry or lactating cows, an investigation can be targeted at the most appropriate area (EL or EDP).
Nutrition affects faecal consistency, and can have an indirect impact on cow cleanliness. While scoring for cow cleanliness, observers should also assess faecal consistency – if excessive variation is present (more than 25% of pats being loose), then a thorough review of nutrition is recommended.
The Mastitis Control Plan recommends that all dairy diets are created by a qualified nutritionist. However, vets do not need to be nutritionists to make a basic assessment of the diet.
Feeding a total mixed ration (TMR) results in more consistent faecal score than feeding roughage and concentrates separately (Schingoethe, 2017). A TMR should be balanced – a ratio of 40% concentrate to 60% forage is commonly recommended, though higher rates of concentrate will be necessary in higher-yielding herds. It’s important to highlight that, with higher concentrate feeding, faecal consistency is likely to be affected (Farm Advisory Service, 2024).
One study found poorer udder hygiene scores in higher-yielding cows (DeVries et al, 2012). Hughes (2001) recommends that crude protein in the TMR should not exceed 18% as this can also affect faecal consistency.
Bacteria, mould or toxins can be present in poorly made silage, or poorly stored concentrate feeds. Unwanted pathogens and toxins present in a diet will disrupt rumen microflora, and can lead to secondary diarrhoea (Fink-Gremmels, 2008). Some toxins can act on the liver or intestine, causing diarrhoea (Smith, 2009). Where bacteria, mould or mycotoxins are identified in the diet the best recommendation is to stop feeding that feed. The next best option, where there is no alternative feed, is to add a mycotoxin binder – calcium clay products have been shown to have some effect where the level of mycotoxin in feed is relatively low (Maki et al, 2016).
Feed availability is also important. Where the feedspace is compromised, or fresh feed is not regularly delivered, cattle are likely to compete at the feedspace, which can affect cow cleanliness. The Mastitis Control Plan recommends that lactating cows have access to at least 75cm per cow, and dry cows have at least 100cm per cow. Fresh feed should be provided daily, with regular feed pushup – at least twice per day in conventionally milked herds, but up to six times per day in automatic milking systems.
Cow cleanliness is an important aspect in controlling environmental mastitis. For lactating cows, this is best assessed in the milking parlour, using an objective scoring system. For dry cows, observers should score far-off and close-up dry cows. In herds with poor cow cleanliness, farmers can make short-term changes to the milking routine, particularly around cleaning cows and pre-milking teat disinfection.
Longer term, the farm should make more important management changes to improve cow cleanliness. This should include attention to space/stocking density, bedding and nutrition. Analysis of clinical mastitis and cell count data can be useful to help in identifying when new infections are occurring. This means that management advice can be targeted where it is most likely to be useful, for example, focusing on the dry cow or lactating cow environment or at particular times of year.
The author (Al Manning) is a trainer for the Mastitis Control Plan (www.mastitiscontrolplan.co.uk/).
The author would like to acknowledge Andrew Bradley and Martin Green, who developed the Mastitis Control Plan with funding from the Milk Development Council, and James Breen and Katharine Leach, who are part of the support team.
Al Manning is an RCVS and European-recognised veterinary specialist in bovine health management. He works at Quality Milk Management Services, a milk recording and mastitis laboratory in Somerset.