7 Apr 2026
Sarah Hunt BVetMed, CertAVP(ECC), MRCVS looks at how the practicalities of recognising this issue quickly can save lives, and the treatment following.

Abdominal ultrasound showing small volume of abdominal free fluid.
Haemoabdomen, also known as haemoperitoneum, is a regularly encountered presentation in emergency and critical care services, and is not uncommonly encountered in general practice.
Rapid recognition and treatment is essential, as unchecked haemorrhage will lead to hypovolaemic shock, anaemia, dehydration and death. Rupture of splenic haemangiosarcoma, where older large breed canines are over-represented, is the most commonly occurring non-traumatic cause.
However, haemoabdomen should be considered in any patient presenting atypically after a recent surgery; where trauma is suspected; or where clinical signs suggest hypovolaemia, anaemia or both.
Ancillary kennel side tests such as non-invasive blood pressure measurement, pulse oximetry, electrocardiography, packed cell volume (PCV), total solids, blood glucose, peripheral lactate, blood gases and pain assessment should be considered during initial triage to establish the degree of compromise and guide initial stabilisation. Arterial blood gas analysis is the gold standard for oxygen assessment, but is not always feasible in the distressed patient; in which case, pulse oximetry and venous blood gases together offer a reasonable alternative initial assessment.
Oxygen supplementation is warranted where pulse oximetry readings for peripheral oxygen saturation percentage is less than 94% or arterial oxygen pressure is less than 90%. Appropriate opiate analgesia should be started if pain is suspected.
Intravenous access should be obtained, and intravenous fluids should be considered if hypovolaemic shock is evident. A hypotensive permissive approach may be justified if ongoing haemorrhage is possible.
Should the degree of anaemic shock be extreme, and not improved after hypovolaemic treatment, then transfusion with compatible packed red blood cells or fresh whole blood will provide much-needed oxygen-carrying capacity to the patient.
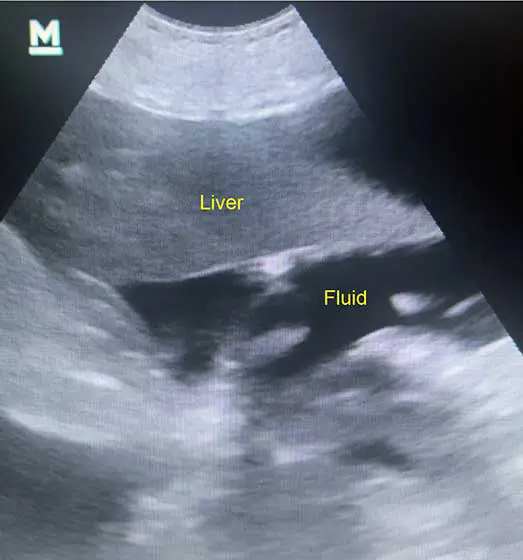
A point-of-care ultrasound, if available, should be readily performed. Free fluid shows as hypoechoic or anechoic regions, with small volumes located most commonly cranial to the liver or caudal to the bladder.
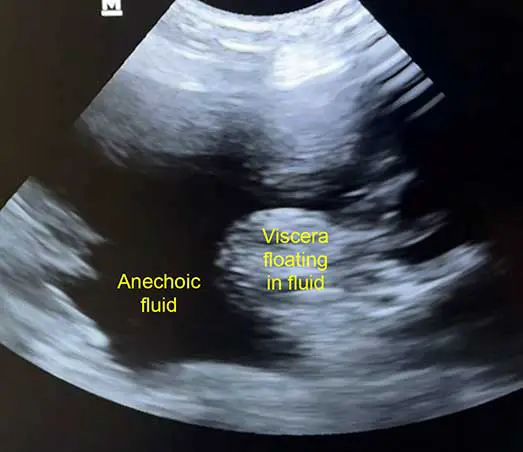
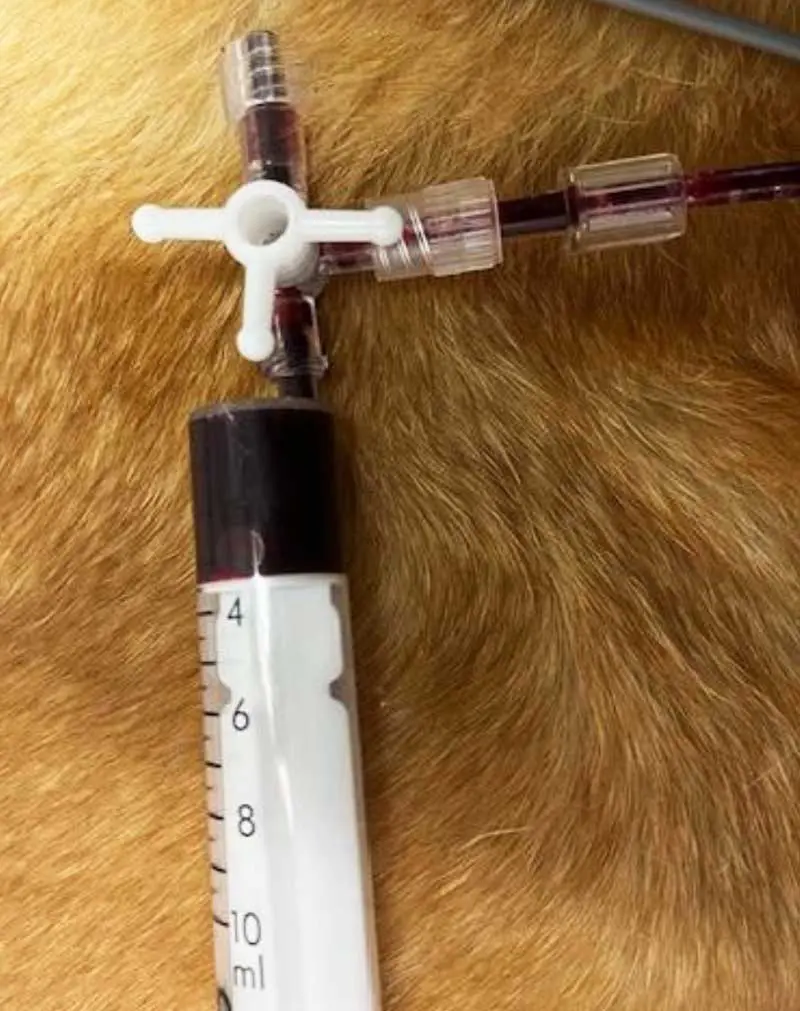
False negatives can occur when the haemorrhage has had the opportunity to clot. Abdominocentesis is a useful tool to differentiate such cases, with very small amounts of haemorrhage detected.
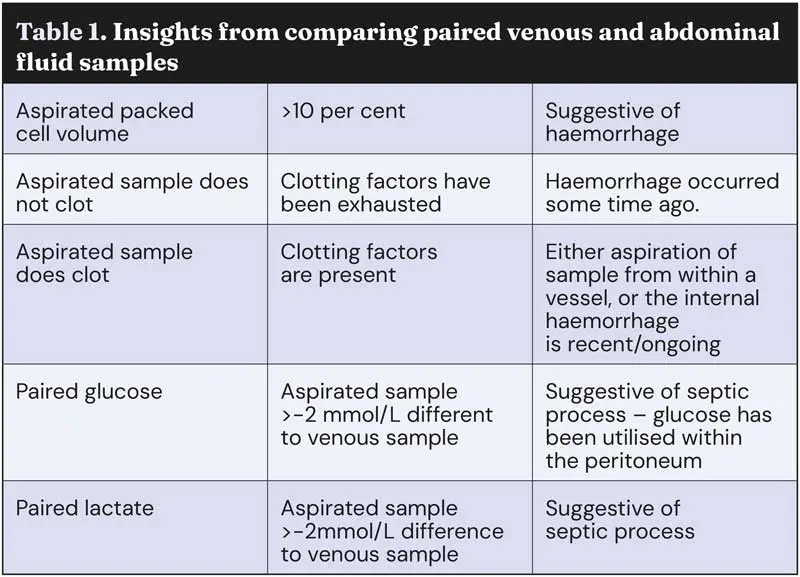
Collected samples should have their PCV and total protein analysed. A PCV of more than 10% is suggestive of haemoabdomen.
Failure of the sample to clot upon exposure to the air indicates chronicity of the bleed, as clotting factors are depleted from the sample. However, if the haemorrhage has occurred less than 45 minutes beforehand, then some clotting ability may be retained.
Where ruptured neoplasia is suspected, an ultrasonic observation of the spleen and heart is recommended, with attention to the right atrium and right auricle, which are common locales for malignancy.
Comparing paired venous and abdominal fluid samples for lactate, glucose and PCV can help characterise the type of effusion, and give insight into cause (Table 1).
Where the haemorrhage could be due to a coagulopathy, such as rodenticide exposure, or is an increased risk of comorbidities exists due to the patient’s age, then a screening serum biochemistry, complete blood count, Angiostrongylus antigen detection test, and clotting functions such as prothrombin time and activated partial thromboplastin clotting time, are sensible screening tests to aid in decision making ahead of treatment.
As the availability of viscoelastic tests becomes more widespread, one would anticipate their widespread incorporation into patient assessment.
If the haemorrhage is deemed to have ceased, and be neither of neoplastic nor traumatic origin, then medical management can be cautiously considered.
Should the haemoabdomen have occurred in relation to a surgery or trauma, then rapid surgical exploration should be encouraged, even without evidence of current ongoing haemorrhage; one must assume that large vessel haemostasis may be disrupted by movement or increases in blood pressure, with potentially catastrophic consequences.
Management of the neoplastic origin haemoabdomen will vary upon individual circumstances, but may be surgical or medically based and either therapeutic or knowingly palliative.
PCV changes are an unreliable and late indicator of ongoing haemorrhage owing to the lag in decreased values following a haemorrhage event, splenic contracture and the influence of intravenous fluid support.
Increasing serial measurements of abdominal fluid pockets by ultrasound gives rapid tangible evidence of ongoing haemorrhage and is more reliable and reproducibly assessed across clinicians than patient mental acuity, PCV changes or hypovolaemic shock clinical parameters.
Depending on the severity of hypovolaemia and anaemia, these patients present an increased risk of mortality during anaesthesia, While efforts should be made to stabilise the patient ahead of surgery, it must be appreciated that ongoing haemorrhage offers the greatest risk to life.
Anaesthetic drugs, doses and speed of administration should be chosen with a view to maintain normotension (Otero, 2013).
Tranexamic acid (intravenous 10mg/kg) can promote clot stability and should be clinically considered in all cases of haemoabdomen, (Kelley et al, 2022), unless an increased risk of thromboembolism is deemed (Moser et al, 1991). Tranexamic acid does not have a current UK veterinary licence, so its use would represent a step four on the veterinary medicines cascade, (VMD, 2015).
Autologous transfusion using cell salvage techniques of the abdominal fluid should be decided ahead of surgery, as collection is best performed at the start of the exploratory celiotomy. The delay in achieving conclusive haemostasis, so as to collect the effusion for cell salvage, should be weighed against the benefits of an autologous transfusion.
Autologous transfusion is generally contraindicated for neoplastic conditions due to the risk of metastatic spread. One could argue that if evidence of metastatic spread is already present, then the risk has been mitigated. Each case warrants an individual risk to benefit consideration by the clinician.
Standard and laparotomy swabs with radiopaque markers should be available, and counted at the start of the exploratory celiotomy.
Suction machines aid visualisation of the surgical field, and when the site of haemorrhage is obvious, artery forceps can be rapidly employed for haemostasis. In postoperative neutering haemorrhage, ovarian or testicular arteries can usually be rapidly located caudal to the pole of each kidney.
In such cases, both sides should be inspected and have an additional transfixing ligature placed, as well as inspection of the cervical stump and broad ligament in females.
In cases of splenic rupture, elevation of the spleen out of the abdominal incision provides gentle traction on supplying blood vessels; this, alongside swab placement over the rupture site, is usually sufficient to prevent catastrophic blood loss while splenectomy is performed.
Where the site of haemorrhage is less apparent, the abdomen should be systematically inspected, using packing swabs to reduce blood entering an area under inspection, then peeling the swab back to look for fresh ooze underneath. Where the rate of ongoing haemorrhage is catastrophic, then aortic cross-clamping can be performed for up to 15 minutes at a time.
Greater cross-clamping durations have been associated with increased morbidity (Akdemir et al, 2014). While surgical haemostasis is always preferable, some haemorrhage sites are not amenable to ligation. In such cases, haemostatic products such as Vetigel (formerly licensed in the UK for dental extractions, now with a more extensive remit) or electrocautery offer general practitioner-level surgeons with haemostatic options.
Once haemostasis is achieved, the abdomen can be closed routinely and hypovolaemia addressed with a more robust fluid strategy.
Where haemostasis cannot be achieved, such as where specialist level surgical skills are required, (for instance, where a liver lobectomy is necessary for definitive haemostasis), then the abdomen should be packed with radiopaque laparotomy swabs, sutured closed to ensure a water tight seal before recovery and urgent transport to a suitable referral centre. The packing and sealing is to increase intra-abdominal pressure around haemorrhaging blood vessels, to reduce blood flow and encourage clot formation.
Sarah Hunt graduated from the RVC in 2005. After a brief stint in mixed general practice, she has spent the vast majority of her career working in out-of-hours services within both referral and general practice clinics. Sarah is an advanced practitioner in emergency and critical care. She is a mum to two school-age children and owns a smallholding with aspirations of food self sufficiency.