5 May 2026
Imaging – innovations and latest thinking for general practitioners
César Sánchez Jiménez DVM, MScVPS, CertAVP(DI), DipECVDI, MRCVS covers radiography, ultrasound and CT as he discusses the role of these modalities in practice.

Figure 1. Thoracic radiographs of a patient presenting with acute dyspnoea.
Since the discovery of x-rays by Wilhelm Conrad Röntgen in 1895, diagnostic imaging has played a fundamental role in the diagnosis and management of disease.
That discovery moved rapidly into veterinary medicine, with reports on veterinary radiology appearing as early as 1896.
More than 125 years later, and despite the development of ultrasound, computed tomography (CT) and magnetic resonance imaging (MRI), conventional radiography remains the workhorse of veterinary imaging.
What has changed in recent years is not simply the availability of equipment, but the way imaging is integrated into clinical workflows. Rising client expectations, wider uptake of pet insurance and the increasing complexity of cases seen in first opinion practice mean that images are expected to inform decision making quickly and more reliably than before.
Advances in digital connectivity, together with the increasing availability of specialist support, now allow practitioners to access expert input through teleradiology, telemedicine and peripatetic services, often within hours of image acquisition or in real time during case assessment. Together with the growing presence of CT in general practice and the emergence of artificial intelligence-assisted (AI) interpretation, these developments are reshaping how imaging is acquired, shared and used in everyday clinical work.
Much of this depends on a system that has been available for years, but is still underused in some practices: picture archiving and communication systems (PACS). More than simple image storage, PACS preserves studies in their original medical format, retaining key diagnostic information such as radiographic attenuation, ultrasound echo characteristics and the spatial references needed for accurate measurements, as well as patient and acquisition information.
Through cloud-based access, studies can be retrieved, reviewed and shared across multiple devices and locations without loss of quality. In practical terms, this means imaging is no longer confined to the people physically present in the practice, but can be discussed, supported and interpreted by radiologists, other specialists and, increasingly, by AI-based tools. In modern practice, PACS should be seen less as optional storage and more as part of the diagnostic process itself.
Radiography: the undervalued tool
Despite the growth of more advanced imaging modalities, radiography remains the first-line diagnostic tool in most small animal practices. Its value is often underestimated, with a persistent perception that it represents “basic” imaging and is straightforward to interpret.
In reality, most specialists would agree that radiography is both highly informative and technically demanding, requiring careful positioning, appropriate exposure and systematic evaluation to extract its full diagnostic value. When used well, it can provide rapid, cost-effective answers in a wide range of clinical scenarios and often reduces the need for immediate escalation to more advanced imaging.
The versatility of radiography is easy to overlook. In Figure 1, thoracic radiographs obtained from a patient presenting with acute dyspnoea showed abdominal organs within the thoracic cavity, with the spleen and small intestine well identified within the thorax on the lateral projection and the colon well visible inside the thoracic cavity on the dorsoventral view. Loss of continuity of the left diaphragmatic margin supported a diagnosis of traumatic diaphragmatic rupture, and the patient proceeded directly to surgery without scalation into further investigation.
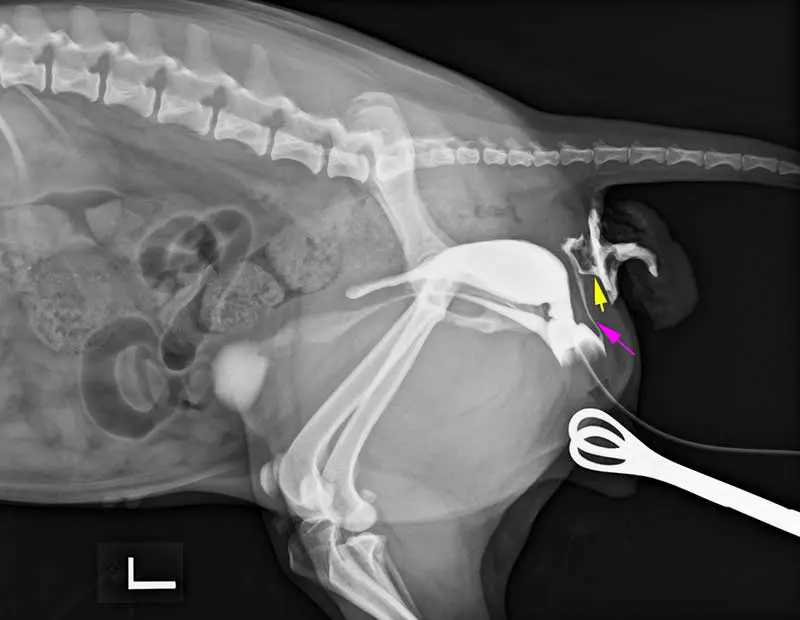
In addition, radiography can be just as valuable when a more tailored technique is used. In Figure 2, a retrograde vaginourethrographic study demonstrates an abnormal communication between the dorsal aspect of the vaginal vestibule (pink arrow) and the mucosa adjacent to the anus (yellow arrow), allowing a definitive diagnosis and more directed treatment planning.
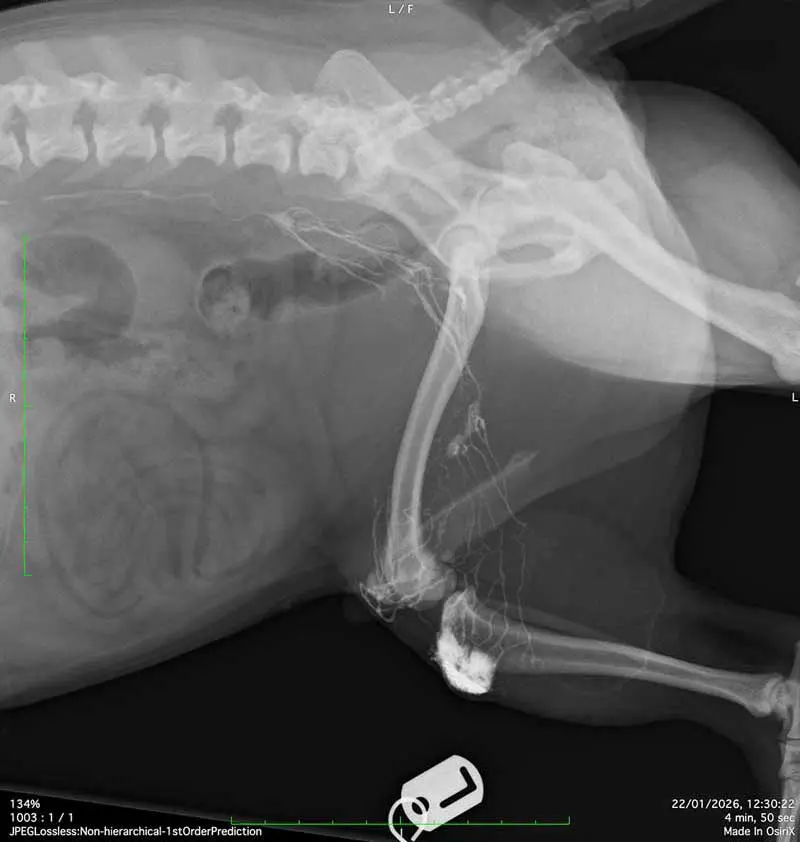
In Figure 3, lymphography performed following intratumoural injection of water-soluble contrast identifies the sentinel lymph node and outlines the draining lymphatic pathway. Together, these cases are a useful reminder that radiography is not limited to survey films; when the appropriate technique is applied, it remains a highly adaptable and often definitive diagnostic tool. Cases like these are being more commonly encountered in veterinary practices, and teleradiology might offer an invaluable aid on quick decision making.
With the widespread adoption of digital imaging, remote interpretation by board-certified radiologists has become an established part of modern veterinary care in many parts of the world, allowing clinicians to obtain specialist input within short timeframes and often at modest cost. Its value, however, depends on more than simply sending images for review. As the American College of Veterinary Radiology and European College of Veterinary Diagnostic Imaging consensus statement on teleradiology makes clear, the quality of the final report is closely linked to the quality of the entire process, including acquisition, positioning, the completeness of the clinical history and the use of consistent quality control.
In this context, teleradiology is best viewed not as a standalone reporting service, but as a structured and collaborative extension of clinical practice.
AI-assisted interpretation is also beginning to find a place in routine workflow, but it should be approached with realism rather than hype. Recent studies suggest that AI is most useful as a triage tool and second reader, rather than as an autonomous diagnostician.
Ndiaye et al (2025) found that commercial AI could perform broadly on par with the best radiologist for descriptive findings and exceeded the median reader in confirming normality, but it was less sensitive for lesion identification and did not generate accurate differential diagnoses.
Other work suggests that AI may be particularly useful earlier in the workflow; for example, in identifying technical problems such as underexposure, rotation or limb malpositioning before studies are submitted for interpretation.
At the same time, external validation work by Ma et al (2026) showed variable performance and frequent missed findings across commercial systems when abdominal radiographs from general practice were assessed, underlining that these tools are not ready for autonomous clinical use. At present, AI is best seen as support for clinical judgement, not a substitute for it.
Ultrasound: the bridge between general practice and referral care
Ultrasound occupies a distinctive position in small animal practice, often sitting between first opinion investigation and referral-level imaging. It is now widely available and increasingly relied upon, not only for abdominal assessment, but also for thoracic, cervical, musculoskeletal and other superficial soft tissue applications.
Its advantages are clear: it is non-invasive, provides real-time information, allows image-guided sampling and can answer important clinical questions rapidly. However, its main limitation remains unchanged. Unlike radiography, ultrasound is highly operator dependent, and the diagnostic value of the study depends as much on image acquisition as on interpretation.
This also helps explain why ultrasound remains such an important modality in first opinion practice. Compared with more advanced imaging, it is often more accessible, more affordable and easier to integrate into day to day case management, while still providing real-time information and the option of image-guided sampling.
Peripatetic imaging services can extend that access further by bringing experienced ultrasonographers into the practice setting, complementing referral care.
Developments already used in human medicine suggest how this model could evolve further. A 2025 health technology review found that asynchronous tele-ultrasound pathways based on standardised acquisition protocols can extend access while maintaining acceptable diagnostic performance in selected applications.
Earlier work by Parsai et al (2012) also showed that standardised video clips improved the diagnostic accuracy of remote sonographic interpretation compared with still images alone. The message is straightforward: if ultrasound is to become more shareable and reproducible, standardisation of acquisition will be just as important as access to expert interpretation.
If these approaches become more established, their value is likely to extend well beyond routine abdominal scanning. Areas that are already used, but still variably adopted in general practice, such as musculoskeletal, cervical and other small-parts ultrasonography, as well as ultrasound-guided procedures, would be particularly well placed to benefit from more standardised acquisition and easier access to specialist review. In this context, the value of procedures such as musculoskeletal ultrasound, that remains a relatively underused application in small animal practice, would become increasingly clear. Musculoskeletal ultrasound is particularly useful for the assessment of tendons, ligaments, muscles and periarticular soft tissues – especially in patients with persistent lameness and limited radiographic findings.
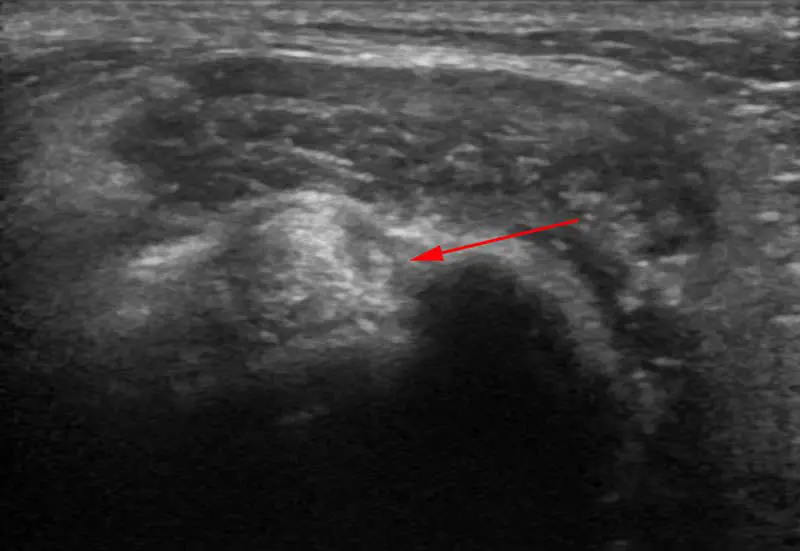
One of its main advantages is that it allows dynamic assessment and can sometimes be performed without sedation, making it well suited to selected orthopaedic cases. In the case shown in Figure 4, a previous CT performed for suspected elbow pathology identified only mild asymmetry of the biceps tendon. Follow-up ultrasound demonstrated tendon heterogeneity together with increased fluid in the tendon sheath, supporting a diagnosis of chronic biceps tendinopathy and helping guide management.
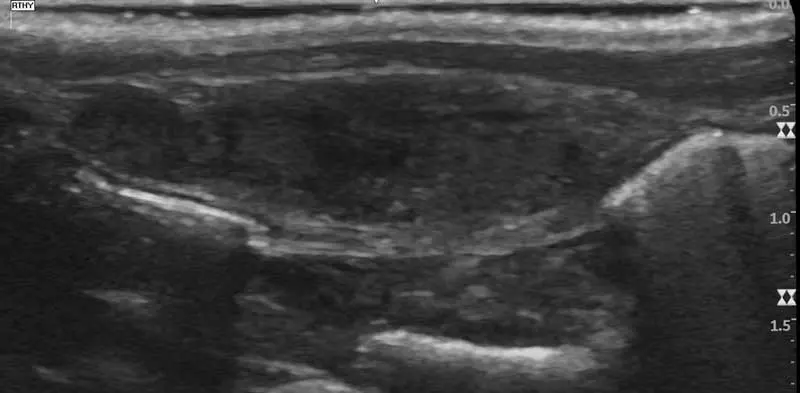
Neck and small-parts ultrasound is another area with considerable clinical value, but one that remains underused in many practices. With high-frequency probes, ultrasonography can provide excellent detail of superficial structures such as the thyroid and parathyroid glands, salivary glands and cervical lymph nodes.
In another example of imaging-guided investigation, a patient presented with reduced appetite and intermittent vomiting rather than the more familiar picture of hyperthyroidism. A cervical scan identified an enlarged and heterogeneous right thyroid gland (Figure 5); further analysis subsequently confirmed a hormonally active lesion in the thyroid and concomitant gastropathy. After this, the patient responded well to medical management.
Ultrasound is also becoming increasingly useful as a procedural and functional tool, extending its role beyond descriptive imaging. This includes guidance for procedures such as biopsy of bladder wall lesions, assessment of ureteral patency and the use of contrast-enhanced ultrasonography to assess organ or lesional perfusion. Other advanced techniques, such as Doppler-derived pulsatility or resistive indices and elastography for tissue stiffness assessment, are not yet part of routine practice in most settings, but they illustrate how the modality is evolving. As expertise and equipment improve, these applications are likely to become more relevant in selected cases.
CT in general practice: with greater access comes greater responsibility
CT is no longer exclusive to referral hospitals. Faster multi-slice systems, shorter acquisition times and wider investment in advanced imaging have made it increasingly accessible, and some general practices now have CT units on site.
That shift has changed expectations from both clinicians and clients: cases that might once have been referred earlier are now, in selected settings, being worked up in house.
The opportunity is considerable, but so is the responsibility to use the modality appropriately, with suitable case selection, protocols and reporting.
It is important to know the technology and, in this instance, cone-beam CT, present in some practices, has a useful but relatively narrow role. In veterinary medicine, it is most established in orthopaedic, dental and maxillofacial imaging, where 3D assessment of dentoalveolar and small bony structures can be extremely helpful – particularly in smaller patients.
Its limitation is that it should not be viewed as interchangeable with conventional CT.
The main value of conventional CT is its superior soft tissue contrast; compared to radiographs, it removes superimposition and allows true cross-sectional assessment. This is particularly important in areas where radiography is limited by complex anatomy, such as the nasal cavity and skull, in thoracic staging, and in selected orthopaedic or spinal cases.
CT can define lesion extent more accurately, detect pulmonary metastases at a smaller size than radiography, and characterise deep structures that are only partly visible on plain films. For the general practitioner, the question is, therefore, not whether CT is better than radiography, but when it adds enough information to justify chemical restraint, contrast and cost.
Where CT can still be underused in practice is in the use of intravenous contrast. Contrast improves lesion characterisation by highlighting vascularity, margins and internal architecture, and for many oncological or inflammatory questions a non-contrast study is only part of the examination.
In practical terms, CT without contrast is often incomplete. Contrast use also requires the same discipline as the scan itself: most patients still need anaesthesia or heavy sedation, intravenous access must be reliable, and patients should be monitored appropriately. Contrast safety is, therefore, part of CT planning, not an afterthought.
Conclusion: imaging in general practice, better connected, better used
Diagnostic imaging in general practice has never been more accessible, more connected or more capable than it is today.
Radiography remains the foundation, ultrasound continues to bridge the gap between first opinion and referral care, and CT is increasingly moving into settings that would once have regarded it as out of reach.
At the same time, PACS, teleradiology, peripatetic services and AI-assisted tools are changing not only how images are acquired, but how they are shared, reviewed and integrated into case management.
The real advance, however, is not simply that practices have more imaging options, but that these options can now be used more efficiently, more collaboratively and, in many cases, with greater diagnostic confidence.
For the general practitioner, the challenge is, therefore, not to use more imaging, but to use the right imaging at the right time and in the right way.
Choosing the appropriate modality, acquiring the study properly and knowing when specialist input is needed remain central to good clinical practice.
Imaging should support clinical reasoning, not replace it, and access to increasingly sophisticated technology does not lessen the need for sound judgement.
Modern imaging is at its most valuable not when it is treated as a standalone test, but when it is used as an integrated extension of the clinician’s thinking.
- This article appeared in Vet Times (5 May 2026), Volume 56, Issue 18, Pages 6-10
César Sánchez Jiménez is a European specialist in veterinary diagnostic imaging. He works across first opinion and referral practices, providing specialist imaging services. César is the founder of VIMOS, a company delivering mobile imaging and teleradiology solutions to veterinary practices.
References
- Banzato T et al (2023). An AI-based algorithm for the automatic evaluation of image quality in canine thoracic radiographs, Sci Rep 13(1): 17024.
- Burti S et al (2024). Artificial intelligence in veterinary diagnostic imaging: perspectives and limitations, Res Vet Sci 175: 105317.
- Canadian Agency for Drugs and Technologies in Health (2025). Asynchronous teleultrasound and in-person ultrasound: comparing diagnostic accuracy, Can J Health Tech, tinyurl.com/55ckv5bp
- Ma D et al (2026). Pilot study: External validation of commercial veterinary radiology artificial intelligence services shows deficiencies in interpretation of general practice–sourced canine abdominal radiographs, J Am Vet Med Assoc 20: 1-8.
- Ndiaye YS et al (2025). Comparison of radiological interpretation made by veterinary radiologists and state-of-the-art commercial AI software for canine and feline radiographic studies, Front Vet Sci 12: 1502790.
- Parsai A et al (2012). Remote sonographic interpretation: comparison of standardized video clips to still images, J Clin Ultrasound 40(8): 495–501.
- Schnelle GB (1968). The history of veterinary radiology, Vet Radiol 9(1): 5-10.
- Ziemer LS et al (2024). ACVR and ECVDI consensus statement for teleradiology, Veterinary Radiol Ultrasound 65(3): 288–293.
