3 Mar 2026
Bryn Jones BVM, BVS, CertAVP, DipECVIM-CA, MRCVS continues his review (first part VT55.42) on research around the pathophysiology, complications and management of the disease.

Figure 1. Mechanism of neutrophils adherence/migration and mechanism of action of fuzapladib. Image: Margaret Dreistadt
Pancreatitis remains a commonly encountered disease in canine practice, providing challenges for both recognition and management. Specific causes, and reasons why some dogs are affected and others are not, are insufficiently understood. Additionally, we do not possess any 100% accurate tests for canine acute pancreatitis (CAP), and severity can vary from self-limiting signs managed at home, to death. For these reasons, research continues into understanding the disease better to allow for possible prevention and intervention.
In the first part of this article (VT55.42), we began looking at the evidence and opinions produced in the past five to six years surrounding CAP. This exploration is continued here with further discussion around causes, complications, diagnosis and management of the disease.
CAP seemingly develops locally within the pancreas, followed by extension to the surrounding tissues followed not infrequently by a systemic inflammatory response (SIRS, which can affect distant organs). Pancreatic secretions are decreased1.
The inflammatory cascade is initiated by activation of NF-κB, which triggers cytokine release and recruitment of neutrophils and macrophages.
Neutrophil migration is facilitated by the leukocyte function-associated antigen-type 1 (LFA-1) domain on the cell, which binds to the glycoprotein intercellular adhesion molecule 1 (ICAM-1) found on many cells, including endothelial cells and leukocytes2.
This binding causes the neutrophils to stop, adhere and then migrate (Figure 1).
Most dogs with pancreatitis are still considered to have idiopathic disease. However, “cryptogenic” may be a more accurate term because our understanding of potential underlying causes is lacking3. Humans can also commonly suffer from idiopathic pancreatitis, though it is less common that cholelith or alcohol-associated disease.
In the previous article, we discussed postoperative pancreatitis, associated endocrinopathies, possible risk of exogenous glucocorticoids (considered low likelihood) and bee envenomation. A recent review, looking at older and newer evidence, provides a good overview of the current opinion regarding causes3.
High-fat diets may worsen pancreatitis in dogs and their potential role in development of pancreatitis have long been suspected and investigated. Diets with less than 20% fat of metabolisable energy have been considered as “low fat”4.
Historical evidence exists for some association of CAP with obesity and feeding of unusual food items/scraps, though any evidence for the role of dietary fat is considered mixed or weak3. Feeding healthy dogs a high-fat diet does not affect cPLI measurements, though this does not faithfully represent the clinical scenario of acute indiscretion of a large fat bolus, and significant conclusions about the aetiology of pancreatitis are difficult to draw5.
Lipid disorders have been suggested as a risk factor for pancreatitis, as excessive fatty acids can be toxic to acinar cells3. Most CAP patients have normal serum cholesterol and triglyceride measurements, and any increases are usually mild6. However, in a small population of dogs with pancreatitis, hypertriglyceridaemia was more prevalent than in healthy controls (18% versus 7.5%), as was hypercholesterolaemia (24% versus 1.9%), though the overall prevalence was still low6. The prevalence of hypertriglyceridaemia increases to 33% for miniature schnauzers with CAP7.
Gianesi et al found that 12.6% of dogs with immune-mediated haemolytic anaemia (IMHA) have suspected CAP at presentation, rising to 22.1% during hospitalisation8. The findings were highly suggestive that CAP is a complication rather than cause of IMHA due to SIRS, ischaemic injury and/or hypercoagulability. Experimental evidence shows that heart failure could exacerbate pancreatitis through reduced blood flow9,10.
Pancreatitis was the most reported complication of treatment in dogs receiving immunosuppressants for precursor-targeted immune-mediated anaemia, affecting four out of 27 dogs (fatal in three), potentially due to their disease or their immunosuppressive therapy (ciclosporin and mycophenolate)11.
In almost 70% of dogs with chronic biliary tract disease, pancreatopathy was also present (44% subclinical and 24% clinical, more commonly chronic), though the exact interplay between the two organs is uncertain12. Pancreatitis was present in 10% of dogs with zygomatic sialadenitis, a condition often association with concurrent disease13.
In dogs receiving mycophenolate for meningoencephalitis, 8.1% developed pancreatitis (distinction between acute and chronic cases is unclear), believed to be a direct result of the medication14.
CAP was reported shortly after administration of an anthelmintic (praziquantel/pyrantel/febantel)15. However, the dog in question had increased liver enzyme and lipase activities prior to administration, making it exceedingly possible that developing pancreatitis was already present.
Due to the severity of inflammation in CAP, damage to surrounding structures is frequently a significant consideration. Often, CAP can show some degree of SIRS, too – not wholly unexpected when circulating digestive enzymes are present. Recognition of these complications is crucial to effective management and recovery, as well as predicting outcome and length or intensity of treatment required. Previously, we analysed pancreatitis-associated kidney injury and extrahepatic biliary duct obstruction.
Acute pancreatitis and systemic inflammation are associated with hypercoagulability in people, and evidence from a small study showed that this is the case in up to 100% of dogs, too16,17. This likely contributes to morbidity and mortality through multi-organ dysfunction and thrombosis.
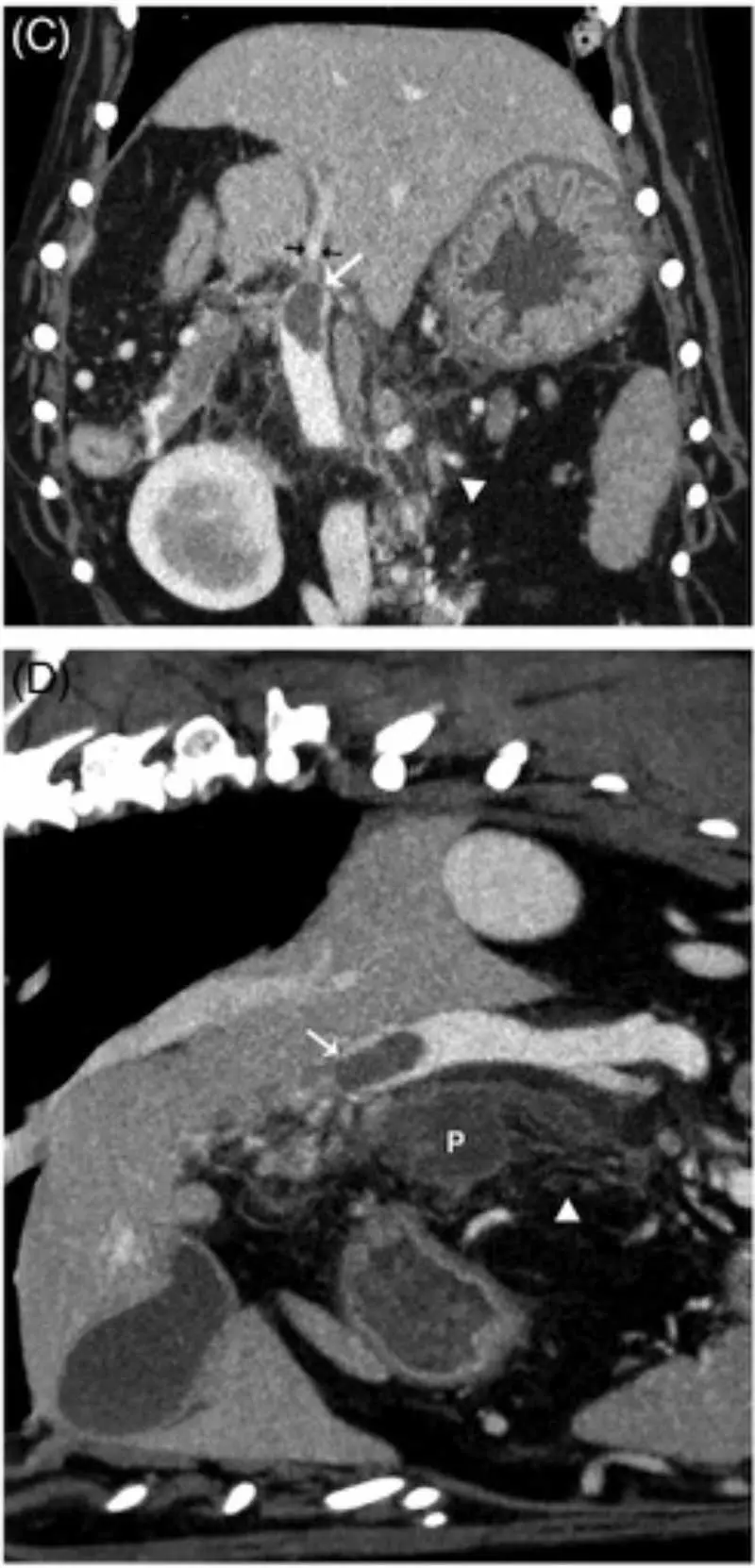
The prevalence of portal vein thrombosis (PVT) was 10 out of 26 and two out of 10 in two CT studies of CAP (Figure 2), and appears associated with increased hospitalisation time, necrosis and mortality18,19. This may be the result of coagulopathy, systemic/regional inflammation or endothelial injury given the proximity. Indeed, pancreatitis is by far the most associated cause of PVT (44%)20,21. Aortic thromboembolism has been reported in at least one case of CAP16.
Clinicians should be alert to the potential for thromboembolic complications, though whether all affected dogs or an identifiable subpopulation may benefit from thromboprophylaxis is an obvious target for future research. Glucocorticoids, colloids and blood products all may increase risk.
Respiratory complications may be more common than previously appreciated, with 13 out of 26 dogs in one study showing respiratory distress on initial assessment, which was associated with increased risk of death22; 21 out of 26 dogs had alveolar or interstitial abnormalities on thoracic radiography.
Although some cases may be due to aspiration pneumonia or thromboembolism, more commonly it is suspected that acute lung injury (ALI) is the cause22. Hyperventilation or tachypnoea can also be a normal response to pain or nausea.
ALIs are considered the most important cause of early death in humans with acute pancreatitis. Diagnosis of ALI is based on specific criteria including acute-onset dyspnoea in dogs with known risk factors (for example, CAP), diffuse bilateral pulmonary infiltrates, and evidence of impaired gas exchange on blood gas analysis. ALI occurs when the pulmonary parenchyma becomes damaged due to marked SIRS and circulating digestive enzymes, with resultant epithelial/endothelial injury. Treatment is predominantly supportive, though development of severe ALI likely implies a guarded prognosis.
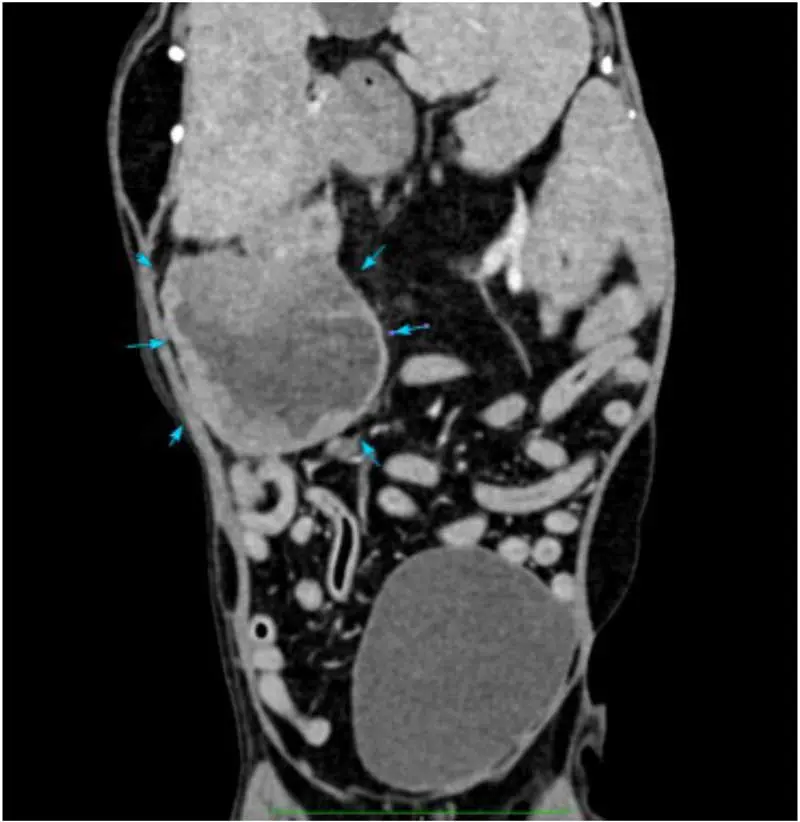
In a minority of dogs with pancreatitis (some acute), pancreatic fluid accumulations can develop (Figure 3)23. These appear to contain neutrophilic inflammation, though the majority have negative bacterial cultures – complicated often by previous administration of antibiotics – so it is unclear how many are septic and how many are a response to severe inflammation.
Medical management can be successful, even leading to resolution, but surgical exploration is ultimately required in some, albeit with a high mortality rate23.
A key limitation in our diagnosis of pancreatitis is the lack of a clear gold standard for comparison. Although histopathology has often been described as the ultimate technique, whether this is truly the best method of identifying clinical pancreatitis is unclear. In one study, all eight dogs with acute pancreatitis categorised as “mild” histopathologically had a final clinical diagnosis that was not CAP24. Biopsy is neither practical nor recommended for most dogs with pancreatitis.
For the tests that are available, they may not be instant (specific lipases or cPLI) or sometimes require experience due to subtlety (imaging), and are still far from as accurate as we would like. Even more frustratingly, almost every study into CAP uses differing inclusion criteria and is, therefore, looking at a different population.
Determining concrete sensitivities/specificity of any individual test may not be applicable to all cases. Some studies have included expert or specialist consensus to come to a conclusion – this is obviously not practical in clinical practice, but highlights the importance involved in interpreting all the results available (“building the picture”)25. In one study, agreement between five internists was good26.
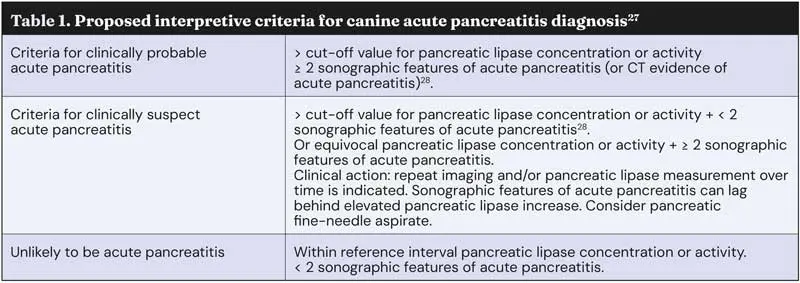
Overall, the favoured approach seems clinician and centre dependent. An interesting alternative would be algorithms that can categorise cases as “clinically suspected” or “clinically probable” pancreatitis (Table 1), as we have for consensus guidelines in other diseases29.
Another novel approach is to use a weighted scoring system involving clinical findings and certain abdominal ultrasonography findings, with a greater emphasis on some more than others (for example, hyperechoic mesenteric fat, hypoechoic pancreas or irregular pancreas)30.
In the previous article, we discussed diagnosis of pancreatitis particularly in relation to ultrasonography; here, we will discuss the use of CT, as well as new opinions around the best use of lipase activities.
A full and thorough discussion of the utility and limitations of lipase activity measurement could fill several articles, and indeed many up-to-date resources exist that discuss this further27,28,31,32. Suffice to say, the sensitivity and specificity of any of these tests is debated, and depends on the diagnostic criteria used.
Many authors (including this one) are potentially happy to diagnose CAP based on clinical signs coupled with ultrasonographic findings, regardless of the results of lipase testing33. Even within the past five to six years, dozens of new papers and reviews have been added to the body of evidence.
Dogs with CAP certainly have higher specific canine pancreatic lipase activity (Spec cPL) than healthy dogs, but overlap between the groups still exists34. A cut-off of 400U/L for Spec cPL has typically been used for diagnosis, with an equivocal zone of 200U/L to 400U/L. Others have suggested a lower cut-off of 350U/L or even 235U/L may be more appropriate35,36. When basing clinical diagnosis on a holistic assessment (including presentation, imaging and lipase testing), sensitivity of cPL for diagnosis of CAP has been reported as 56% and specificity of 86% using the 400U/L cut-off, and only slightly higher sensitivity of 68% using 200U/L36. Spec cPL may correlate with the clinical severity of disease, but this is not a universal conclusion37.
Ultrasonographic findings only weakly correlated with Spec cPL in one study of dogs with acute gastrointestinal signs38. Spec cPL seems to reduce or normalise faster than ultrasonographic abnormalities in CAP35,39. Chronic glucocorticoid use appears to increase Spec cPL and 1,2-o-dilauryl-rac-glycero-3-glutaric acid-(6’-methylresorufin) ester (DGGR) lipase activities, though the clinical significance is uncertain40-42.
While point-of-care cPL tests have traditionally been considered extremely sensitive and less specific, a recent study found a sensitivity of 83% and a much higher specificity (95%)26. Of some interest, presence of an obstructive gastrointestinal foreign body alone does not appear to induce false positives on Snap cPLI testing1,43.
DGGR lipase activity measurement has been proposed as a quicker and more readily available method with similar performance36,44. As for Spec cPL, the optimal cut-off values are probably still uncertain44,45. Specificity is not perfect – increased DGGR lipase was frequent and often marked in dogs with acute kidney injury, likely due to reduced clearance, even though only a small number had suspected pancreatitis46. In a study of critically ill dogs, only 32% of the dogs with increased DGGR lipase at admission had a clinical diagnosis of CAP, with other causes including immune-mediated disease, renal disease and airway obstruction, emphasising the importance of good patient/population selection and application of testing42. This may be because DGGR lipase does not appear to necessarily be pancreas specific30.
Hepatopathy alone does not appear to induce changes in DGGR lipase activity, though portal hypertension might do47,48. Hyperadrenocorticism may have some link with pancreatitis in dogs (see part one), but DGGR lipase activity does not appear to be accurate for diagnosis of CAP in these patients – increased activity (more than 118U/L in that study) was present in 64% of dogs, with hypercortisolism lacking ultrasonographic evidence of pancreatitis49.
With the increasing availability of CT, more dogs are likely being diagnosed in this way (Figure 4). Although AUS has traditionally been the main method, and indeed changes are often part of the inclusion criteria for studies, CT is considered the gold standard in diagnosing human pancreatitis50. An advantage of CT is that the entirety of the pancreas can be reliably visualised, which is possible in only three-quarters of dogs via ultrasonography, even by specialists18.
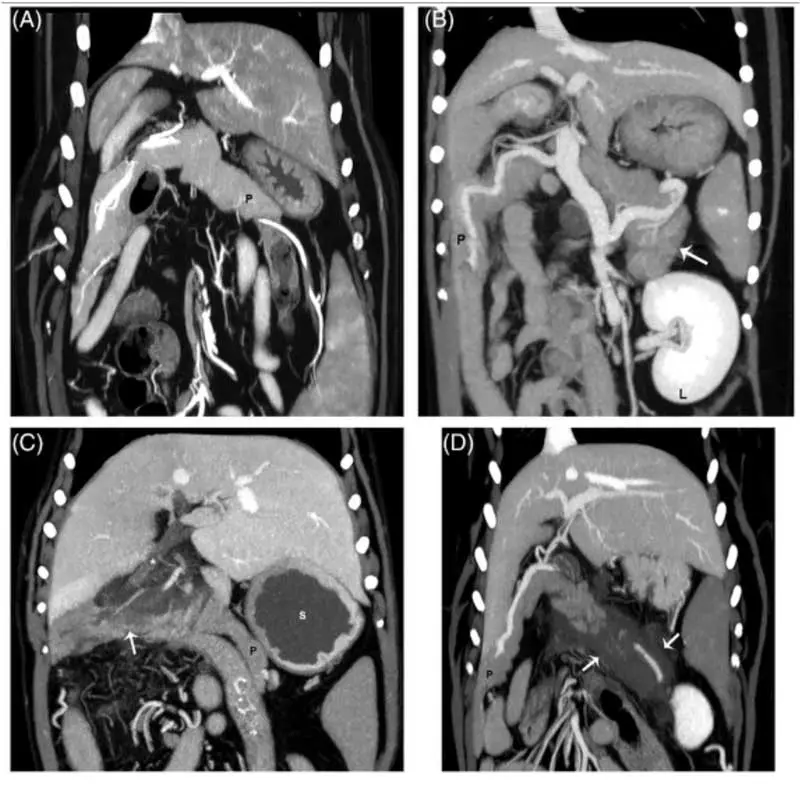
Heterogenous pancreatic contrast enhancement in people is a signifier of necrosis, a significant prognostic marker (some older evidence suggests this in dogs, too), and CT has been shown as a very sensitive test for this in dogs18. CT is superior to ultrasonography for identifying portal vein thrombosis (present in 10 out of 25 cases; Figure 2), which does have important prognostic importance18,20,21. However, despite all these advantages, CT has not yet been found to be superior to specialist ultrasonography for the actual diagnosis of CAP (for example, pancreatomegaly or mesenteric changes)18.
As discussed in part one, pancreatitis in dogs once diagnosed is generally treated supportively and symptomatically, and we previously looked at the potential benefit of glucocorticoids.
Use of anti-emetics (for example, maropitant, metoclopramide or ondansetron) is common to control signs and improve appetite. Many severely affected dogs can require hospitalisation for fluid therapy and effective analgesia. Recent human evidence suggests that use of lactated Ringer’s solution can reduce progression of pancreatitis compared to saline51. For refractory pain, the use of an ultrasound-guided erector spinae plane block has been described and could be an option for further exploration52.
Enteral nutrition is a top priority, which may necessitate the placement of feeding tubes, and early feeding is now recommended53. Withholding food may promote enterocyte damage and dysbiosis. While low-fat diets are typically recommended for dogs with chronic pancreatitis, their importance in CAP is probably much lower. All things considered, lower fat diets are probably a good option if possible, even if simply to encourage gastric emptying1,54,55.
In human pancreatitis, exogenous pancreatic lipase is occasionally administered to combat temporary pancreatic insufficiency due to obstruction/disruption of normal secretory function. Their use is not currently widespread in veterinary medicine, but very unlikely to be harmful (if they do not dissuade eating) and are certainly a consideration in dogs struggling to maintain weight or with diarrhoea.
Fuzapladib is an intravenous daily drug that has been studied for its potential therapeutic effects in CAP and is licensed in other countries2. Fuzapladib prevents LFA-1 from moving to a high affinity state and binding to ICAM-1 (Figure 1) and, therefore, prevent circulating neutrophils from adhering to the endothelium and migrating into tissues, theoretically reducing systemic inflammation.
The study into the effectiveness of this medication excluded dogs with more severe disease and multi-organ involvement, and based diagnosis solely on presenting signs and increased cPLI activity, opening the door for some false positives.
Out of 61 treated dogs, no dogs that died were clearly attributable to the treatment itself, though some severe outcomes were reported. After three days of treatment, one clinical severity score was significantly lower in the treated versus placebo group (92% versus 68% decrease in score). Some criticism of the study included its exclusion of more severe CAP, that some dogs were treated as outpatients without “standard” therapy, the less-strict inclusion criteria, and the potential for the low number of severe adverse effects to be related to the treatment56.
The authors acknowledged some of these weaknesses, but highlighted that exclusion of more severe cases was more likely to underestimate the benefit, and that the drug’s main advantage was in preventing progression of disease form mild to severe forms rather than treating severe disease57. Fuzapladib (Panoquell-CA1) is conditionally licensed in the US and fully licensed in Japan, opening the door for clinical use and hopefully further, more robust data to come. This medication is not currently available in Europe, but is clearly something to remain aware of for the future.
CAP can be a severely debilitating and even fatal disease for some dogs (11% to 46% mortality rate reported, though heavily biased towards dogs hospitalised at referral centres)16-18,22,58-66.
Many dogs can go on to develop chronic or recurrent disease even after recovery, and attempts to identify those patients is laudable, as it may allow setting of expectations or even prevention.
Bryn Jones qualified from the University of Nottingham in 2014 and moved into small animal general practice in south Wales for four years, where he completed a CertAVP. After completing a small animal medicine residency in Hampshire, he moved to Willows Referral Service in 2023, where he currently works. He has earned recognition as an RCVS and European specialist in small animal internal medicine. Bryn has an particular interest in hepatology, nephrology and especially gastroenterology.