24 Feb 2026
Lucy Sheard BSc(Hons), BVSc, CertAVP, MRCVS discusses the options available to the UK equine population, to aid in discussions on their justification to owners.

Image: rima15 / Adobe Stock
Vaccinations are a fundamental component of equine preventive health care, serving to protect both the individual and local and national herd. While the core vaccinations against equine influenza virus (EIV) and tetanus are generally well understood by horse owners, awareness and uptake of the vaccinations available for other infectious equine diseases is variable.
This article summarises the vaccinations available and the justifications for vaccination for all life stages in the UK equine population.
EIV is highly infectious and contagious, and affects all equids. Clinical signs typically include a non-productive cough, pyrexia, serous nasal discharge, lethargy and inappetence.
Infection is not typically fatal, although young, immunocompromised or immunologically naive animals may be more severely affected. The virus may be transmitted directly through inhalation of infectious aerosols or indirectly through fomites.
Numerous outbreaks of EIV have occurred around the world, most recently in 2019 at a large scale in Great Britain, as well as smaller more frequent outbreaks, primarily affecting unvaccinated animals (Whitlock et al, 2023).
This disease may have profound consequences for individual animals, but large-scale outbreaks may lead to temporary cessation of equine sport and mass disruption of the equine industry.
The instigation of mandatory vaccination against EIV by several national and international equine sporting regulatory bodies to prevent further outbreaks has led to reasonable uptake by the equine community. Though the exact proportion of horses vaccinated in the UK is unknown, estimates range from 30 per cent to 71.3 per cent, with lower rates reported in non-competition animals (Wilson et al, 2021).
Vaccination aims to reduce the severity of clinical signs and viral shedding. Several products are available to vaccinate against EIV, or EIV and tetanus. Clinicians are directed to the summary of product characteristics (SPC) of the specific product available to them, as well as relevant sporting bodies, for product-specific dosing regimens.
Broadly speaking, the immunisation course may be administered from six months old and consists of an initial IM vaccination. A second dose is then administered between four and six weeks later, with a third dose required five months after this. Subsequent boosters may be administered every 12 months, though some regulatory bodies mandate six-monthly boosters.
Tetanus is the clinical disease caused by toxins produced from Clostridium tetani infection. The bacterium is typically found in soil and gains entry through wounds; therefore, all horses are susceptible to infection, irrespective of their purpose or environment.
Individual vaccination against tetanus is an IM administration of inactivated tetanus toxoid to stimulate active immunity. The primary course consists of an initial dose from six months, followed by another dose four weeks later.
The next vaccination should be no later than 18 months after starting the primary course, and subsequent boosters should be every 24 months after that (MSD Animal Health, 2023).
While nine different equid herpesviruses (EHVs) have been reported, the most clinically relevant are the pathogenic alphaherpesviruses EHV-1 and EHV-4.
Both forms of the virus are respiratory pathogens and may cause rhinopneumonitis. Additionally, EHV-1 can cause abortion, neonatal foal death and equine herpesvirus myeloencephalopathy (EHM).
Outbreaks of EHV-1 and, to a lesser extent, EHV-4 can have significant consequences for horse welfare, as well as financial ramifications for equine sport.
Vaccination primarily aims to reduce the incidence of EHV-1 associated abortion. It is ineffective at preventing development of EHM, and vaccination in the face of an outbreak is not recommended.
The primary vaccination course against EHV-1 and EHV-4 can be administered from five months, with a second injection four to six weeks later. Revaccination should be performed every six months following on from the primary course. It is recommended that all horses used for breeding purposes or those resident on a stud farm be vaccinated against EHV.
West Nile virus (WNV) is an insect vector-borne flavivirus transmitted through mosquitoes. Horses are considered a dead-end host, and while the disease itself is zoonotic, no evidence exists of horse-to-horse nor human-to-horse transmission. Clinical signs include ataxia, fever, muscle fasciculations, limb paresis or paralysis, or other neurological deficits (Paré and Moore, 2018).
Horses may also be subclinically infected. While clinical cases in the UK have previously been limited to horses with recent history of travel outside the country, native cases may soon emerge, as the virus has recently been detected in UK mosquitoes (Santos and Roxby, 2025).
Three vaccinations against WNV are marketed in the UK, although product availability may be limited. These vaccines are designed to reduce viraemia as well as the duration and severity of clinical signs.
The primary course for Equip WNV (Zoetis) and Equilis West Nile (MSD Animal Health) consists of an initial IM vaccination from six months old, followed by a second dose three to five weeks later. Proteq West Nile (Boehringer Ingelheim) may be administered to animals from five months old and should be repeated four to six weeks later.
Subsequent doses for all products should be administered at yearly intervals. Vaccination should be recommended to horses considered at risk, such as those that travel to countries where disease is endemic.
Strangles is another highly contagious respiratory disease and is caused by the bacteria Streptococcus equi subspecies equi. Clinical signs include pyrexia, lethargy, cough, dysphagia, nasal discharge, lymphadenomegaly and abscessation of the lymph nodes.
The disease is spread through direct contact with infected bodily secretions or indirectly through fomites. While S equi has poor environmental survival, disease spread is perpetuated by subclinical carriers.
Previous products for vaccination against S equi have been blighted by limited efficacy, side effects and difficulty in administration (Rendle et al, 2025). Strangvac has recently been marketed by Dechra and consists of recombinant S equi proteins. The use of recombinant proteins not only increases the safety of the vaccine, but also enables the differentiation of infected from vaccinated animals (Rendle et al, 2025).
Vaccination is designed to reduce the severity of clinical signs associated with strangles in the acute stages of infection (Intervacc AB, 2025). The proposed dosing regimen consists of an initial vaccination administered intramuscularly from five months old, and a second dose administered four weeks later. The SPC recommends that the primary course be repeated after two months in high-risk animals; however, a perspectives article by Rendle et al (2025) proposed that longer dosing intervals may be sufficient (Intervacc AB, 2025).
As the product is relatively new to market, published reports of the product’s use, safety and efficacy are limited, though initial reports are promising (Robinson et al, 2020).
Equine viral arteritis (EVA) is a respiratory and reproductive disease caused by equine arteritis virus (EAV).
Horizontal transmission may occur through contact with virus particles in aerosolised bodily secretions, urine, faeces, aborted material or fetal membranes. Venereal transmission may occur through natural cover or artificial breeding with infected semen.
The virus can cause both subclinical infection as well as overt clinical signs, typically presenting as abortion or neonatal death. Stallions that are persistently infected act as a reservoir for transmission within the breeding population (Balasuriya et al, 2018).
Equip Artervac, marketed by Zoetis, is the only vaccination against EAV/EVA in the UK at the time of publication and its use has been limited by inconsistent product availability.
The recommended vaccination schedule is an initial dose from nine months old, followed by a second dose three to six weeks later. Boosters should then be administered at six-monthly intervals.
Vaccinations should only be administered to stallions and teasers, and animals should not be vaccinated less than three weeks prior to breeding.
It is not currently possible to differentiate vaccinated animals from infected animals using serological testing, so testing prior to vaccination in unvaccinated or lapsed animals is necessary to confirm that the animal is seronegative.
Equine Infectious Disease Surveillance has published guidelines and a decision tree to guide best practice in vaccination (Equine Infectious Disease Surveillance, 2025).
Broodmares should be vaccinated against EIV and tetanus prior to conception. Additional vaccination against EIV and tetanus between four and six weeks prior to foaling may confer additional antibody protection to foals through passive transfer.
Mares in foal should be vaccinated against EHV-1 and EHV-4 in the fifth, seventh and ninth months of pregnancy to reduce the risk of abortion. Equine rotavirus is the most common cause of neonatal diarrhoea and may result in clinical disease in foals as young as 12 hours old (Eertink et al, 2024). If left untreated, rotaviral diarrhoea can lead to electrolyte derangements, severe diarrhoea and may ultimately be fatal.
Vaccination of the broodmare in the eighth, ninth and tenth months of pregnancy leads to the passive transfer of maternal antibodies against equine rotavirus A to foals through colostrum, reducing the risk of rotaviral enteritis and diarrhoea. Vaccination does not confer immunity against equine rotavirus B.
Foals that receive adequate passive transfer from vaccinated mares have been demonstrated to have increased antibodies for approximately 60 days (Zoetis Belgium, 2025).
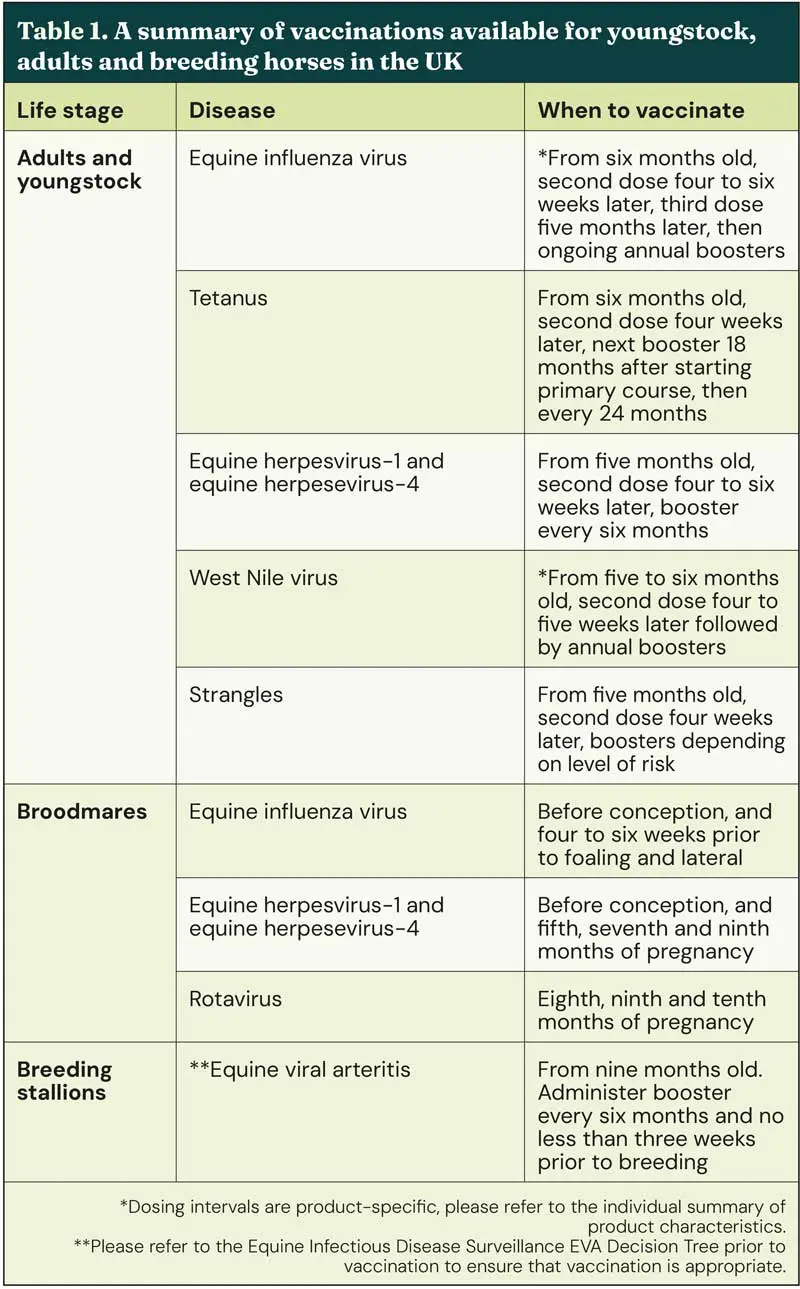
A summary of the vaccinations available can be found in Table 1.
Vaccination should play a fundamental role in equine preventive health care. Veterinarians should educate owners and promote appropriate, risk-based vaccination to promote both individual and herd immunity.
Additional vaccinations are strongly recommended in breeding animals, and immunisation of the mare gives the best chance of passive transfer of antibodies to foals.