24 Feb 2026
Ferran Valls Sanchez DVM, DipECVIM‑CA, MRCVS explains the updated classification of, and process for diagnosing, these gastrointestinal disorders in dogs.

Image: adogslifephoto / Adobe Stock
I wrote this article a few days before the end of last year – a time for social media wrap-ups and lists of goals and wishes. I want to go to Egypt in 2026, and while you are reading this, your mind has inevitably conjured an image of a pyramid.
Today, we will use a pyramid as a framework for this article1,2, albeit, a different one (Figure 1).
On the left side of Figure 1, you can see how chronic inflammatory enteropathies (CIE) were previously classified, and on the right, the updated classification.
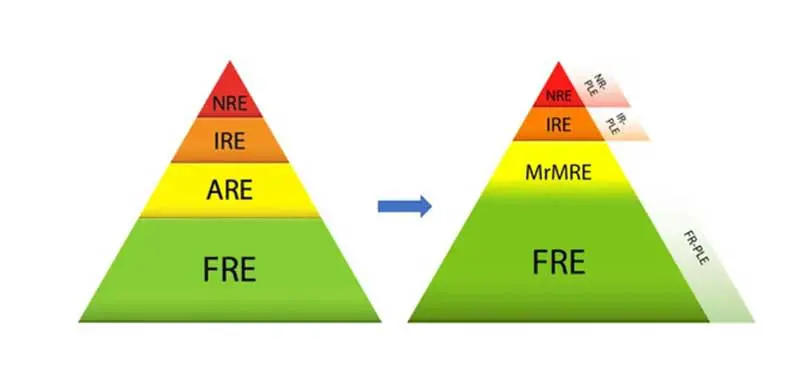
When we suspect that a patient may suffer from CIE, remembering this image is extremely useful, as it reflects which type of CIE is more common.
In addition, it illustrates the pathway to reach a diagnosis: if we understand the pyramid as an arrowhead, it shows in order what to rule out or rule in first, then second, and so on.
CIEs are inflammatory conditions (with histological evidence) of the gastrointestinal tract, characterised by chronic or recurrent gastrointestinal signs such as vomiting, diarrhoea, abdominal pain, nausea, weight loss, or a combination of these. This disease is multifactorial in origin, involving genetics, diet, microbiota and immune system factors2.
Chronic vomiting and diarrhoea are common clinical complaints, and CIE should always be considered when establishing a list of differential diagnoses for these signs. This is particularly true in older dogs, where infectious causes are less likely and CIE and neoplasia are often at the top of the list.
CIEs are classified into food-responsive enteropathy (FRE), microbiota-related modification-responsive enteropathy (MrMRE), immunosuppressant-responsive enteropathy (IRE) and non-responsive enteropathy (NRE). However, I prefer to refer to the last two as inflammatory bowel disease (IBD).
All of these subtypes can lead to a syndrome called protein-losing enteropathy (PLE), in which protein leakage and malabsorption result in severe hypoalbuminaemia. This represents a more severe clinical scenario and should raise greater concern. PLEs are more commonly associated with IBD, but some PLE cases respond exclusively to dietary management, meaning that FRE can also cause PLE.
FRE is diagnosed when gastrointestinal signs resolve or significantly improve within two weeks of starting a diet trial.
FRE is the most common type of CIE, accounting for more than 50% of cases. These dogs are often younger and present with milder clinical signs. Hydrolysed diets or novel protein diets (homemade or commercial) are usually among the first choices. It is important to raise awareness of the relevance of dietary management as a first-line treatment in CIE. In one study, two-thirds of cases initially considered non-responsive to diet responded to a different diet trial, and many treatment-naive cases improved with exclusive dietary management3. Another research group reported similar findings, with 19 out of 24 dogs reclassified from NRE to FRE over a one-year period4.
Setting expectations regarding the number of diet trials a dog (or cat) may require during initial consultations is fundamental. Unpublished data suggest that some patients may need up to eight diet trials before finding an effective one. In my opinion, acceptance of multiple diet trials is higher when this is discussed at the first consultation.
A thorough dietary history should be obtained to understand which diets have been used previously and how they were administered, including owner compliance. Sometimes, I ask pet parents to write down a list of diets used (type, duration, whether other foods were given, and whether any clinical improvement was observed).
Finally, if the dog is reluctant to eat the recommended diets, referral to a nutrition specialist should be offered, as they can formulate a homemade diet tailored to palatability.
In recent years, there has been extensive research on the gut microbiota, with growing evidence supporting the use of probiotics and faecal microbiota transplantation (FMT). Alterations in gut microbiota composition and function (dysbiosis) are associated with canine CIEs1.
As shown in the updated pyramid compared with the older version (Figure 1), there is now a recognised group of cases that respond to microbiota-related interventions. Antibiotic-responsive enteropathy has been removed from the classification.
In dogs with CIE refractory to dietary management, FMT has been shown to improve clinical signs5. Dogs with milder clinical signs and absent or mild dysbiosis tend to respond better. Dysbiosis can be assessed using the dysbiosis index (DI). Another study suggested that FMT may also serve as an adjunctive treatment in dogs with CIE, with better responses again seen in dogs with lower DI values6.
FMT should be strongly considered when diet trials have failed, before moving on to immunosuppressive therapies. One of my earlier mistakes was using FMT as a “last resort” (for example, in severe PLE cases, where it predictably failed). Patient selection is, therefore, crucial to maximise success. In 2024, guidelines were published outlining appropriate FMT protocols7.
Regarding probiotics (live microorganisms), prebiotics (substrates for beneficial microorganisms) and synbiotics (combinations of both), the main conclusion is that further large-scale studies are needed. Some studies using an eight-strain lactic acid bacteria mixture and Saccharomyces boulardii have shown promising results2.
Finally, one publication reported clinical remission in two dogs with non-responsive IBD treated with colestyramine, a bile acid sequestrant8. The proposed mechanism is correction of excess primary bile acids resulting from bile salt dysmetabolism associated with intestinal dysbiosis.
According to several studies, IREs account for approximately 10% to 25% of CIE cases1. Prednisolone is the most commonly used first-line treatment. If there are significant side effects or inadequate response, other drugs such as ciclosporin or chlorambucil may be added.
A thorough discussion of prednisolone side effects is essential, so that pet parents are informed and do not panic. This is also an opportunity to provide practical advice to improve quality of life for both the dog and the owner (for example, increased access to outdoor toileting, constant access to water, caution with leftovers and bins if scavenging develops, and avoiding ad libitum feeding).
As mentioned previously, a careful review of the clinical history and previous diet trials (to confirm that FRE has been adequately excluded) is fundamental before initiating immunosuppressive therapy.
Some cases do not respond to immunosuppressants and are associated with a poorer prognosis.
In the past, antibiotics such as metronidazole were commonly used in cases of chronic diarrhoea. However, over the past decade, evidence has emerged showing limited benefit and significant negative effects on gut microbiota9.
Persistent alterations in microbiota diversity have been reported following the use of metronidazole and tylosin, which may explain the high relapse rates observed after treatment discontinuation9.
Additionally, global concerns regarding antimicrobial resistance make antibiotic stewardship particularly important within the one health framework.
Ferran Valls Sanchez graduated from the Autonomous University of Barcelona in 2011 and is a European specialist in small animal internal medicine and co-founder of WeYouVets, a veterinary startup dedicated to bringing specialist internal medicine support directly to first-opinion practices, alongside high-quality education. Ferran is passionate about making specialist knowledge practical, accessible and relevant to everyday clinical work. In 2025, he also co-founded VetCracks, an educational initiative for veterinarians in Latin America, built on the same values as WeYouVets and focused on accessible, high-quality, and people-centred veterinary education. Visit weyouvets.com