3 Feb 2026
Ian Wright, BVMS, BSc, MSc, MRCVS, considers how the distribution and activity of some key ticks and their pathogens are changing, with a particular focus on the UK

Tick-borne disease is a growing risk to UK pets and their owners, with significant changes in tick activity and the introduction of tick-borne pathogens over the past two decades. These changes are also occurring across mainland Europe, increasing the chances of UK pets encountering ticks abroad and novel pathogens being introduced to the country.
As the distributions of ticks and their pathogens rapidly evolve, it is important that these changes are monitored and incorporated into veterinary care. The distribution of ticks and their pathogens impacts differentials for clinical disease in dogs and cats, and informs tick prevention advice for pets and their owners.
Ixodes species is the most common tick in the UK and widespread across the whole of Europe (Abdullah et al, 2016; Wright et al, 2018).
Traditionally in the UK, spring and autumn peaks in Ixodes species tick activity have been recorded. Real-time data from the Tick Surveillance Scheme (TSS), however, suggests that an extended peak now occurs from early spring though until late autumn, with the highest levels of exposure occurring during May and June. These data also show that while these are higher risk times for UK tick activity, exposure to ticks can occur at any time of year (Wright et al, 2018; Hansford et al, 2023). Therefore, veterinary professionals need to be aware of tick exposure and potential pathogen transmission throughout the year.
Two key pathogens transmitted by Ixodes species ticks with increasing disease incidence and geographic range are Borrelia burgdorferi and tick-borne encephalitis virus (TBEV), respectively.
Most canine infections with Borrelia burgdorferi sensu lato are subclinical, with 5% to 10% of infected dogs going on to develop clinical signs of Lyme disease. These include acute or subacute arthritis in one or more joints, fever, anorexia, lethargy and lymphadenopathy.
Dogs present primarily with the acute form, which is often transient, although relapses can occur. Other immune-mediated signs may also be observed, although the severe form of Lyme nephritis observed in the US has not been recorded in the UK. Chronic disease is not commonly recognised in canines, but has been associated with non-erosive polyarthritis and an increased risk of chronic renal disease (Drake et al, 2018).
Lyme disease is rarely recognised in cats, but it is unknown whether this is due to cats being relatively resistant to exposure, resistant to disease, or whether it is under-reported and under-diagnosed in felines; two suspected cases of heart disease secondary to B burgdorferi have been recorded in the UK, however, so should be considered as a differential in cats with relevant clinical signs (Tørnqvist-Johnsen et al, 2020).
Little information exists on the UK prevalence of B burgdorferi infection in cats and dogs, but is present widely in ticks removed from them. Two large studies have examined the distribution and prevalence of ticks infesting domestic dogs as well as the prevalence of B burgdorferi in these ticks (Smith et al, 2012; Abdullah et al, 2016). The prevalence of Borrelia species found in Ixodes species ticks attached to dogs has only increased marginally, from 2.3% to 2.37%, in the time between the two studies, but the overall number of ticks has increased. This is a picture largely replicated across northern Europe. This suggests that dogs are being exposed to increased numbers of Borrelia species infected ticks, but research is still required to establish how this is translating into the prevalence of Borrelia species infection and incidence of Lyme disease.
A study analysing data from vector-borne pathogen tests submitted to Idexx diagnostic laboratories across Europe between 2016 and 2020 found a seroprevalence for B burgdorferi in UK dogs of 1.1% from 2,631 samples. No information is available on what proportion of these tests were screening tests and which were in clinically unwell animals. If representative, this suggests a small, but significant, proportion of UK dogs are exposed to infection (Miró et al, 2022).
TBEV and Louping ill virus (LIV) are both zoonotic flaviviruses transmitted by Ixodes species ticks.
Historically, TBEV has been endemic in Europe, with its distribution having rapidly expanded this century. Conversely, LIV’s geographical distribution is predominantly in the UK. Recently, however, evidence of TBEV endemic foci have emerged in the UK, complicating the epidemiological picture. Both viruses can cause progressive, and sometimes fatal, neurological signs in dogs. They are also both capable of causing severe zoonotic disease if transmitted to humans via tick bites.
The European subtype of TBEV has rapidly spread across Europe in the past two decades and is now present in Scandinavia, and central, eastern and western Europe, including the UK. A surveillance programme carried out by Public Health England in 2018 looked for evidence of TBEV in wild animals and ticks. Serum was collected from 1,309 deer culled across England and Scotland; 4% of samples were ELISA positive for TBEV, with foci in the New Forest and Thetford Forest. PCR positive ticks were also collected from the Thetford Forest (Holding et al, 2020).
Rodents act as reservoir hosts capable of maintaining the pathogen once it has moved to a new area, and larger wild animals such as deer serve as transport hosts for infected ticks. These transport hosts, as well as migratory birds, are thought to have played a large role in rapidly disseminating the pathogen through abundant Ixodes species populations across Europe. In the UK, it is likely TBEV is present in multiple foci across the UK and is likely to spread.
LIV, in contrast, has a predominant geographical distribution in the UK’s upland areas (Jeffries et al, 2014), with reservoirs in grouse and sheep. This means that large geographic areas of the UK are likely to be harbouring ticks infected with one or both pathogens, with an unknown overall distribution of TBEV. The risk of exposure should be kept in perspective for concerned pet owners, however, as the risk of human and canine infection from short visits to endemic areas with limited tick exposure is thought to be low.
Ixodes species ticks can transmit a number of pathogens that can be carried by dogs, including Borrelia species, TBEV, Anaplasma phagocytophilum and small Babesia species. No evidence exists to suggest, however, that dog or cat ownership is a direct risk for infection, suggesting that pets bringing ticks into households or into direct contact with people is a significant risk factor for human tick exposure.
Positive dogs, though, are useful as sentinels for human infection, and seroprevalence of dogs is a useful sentinel for human seroprevalence for tick-borne pathogens in national populations (Cook and Puri, 2020). Human cases of Lyme disease have increased in the UK human population over the past decade, and a recent paper looking at retrospective GP records on Lyme disease diagnosis suggests that official government figures are a significant underestimate of the true total (Cairns et al, 2019). The growing number of reported cases is likely to be a combination of a genuine increase in pathogen transmission, heightened awareness among the general public and increased surveillance. It is unknown whether a similar increase is occurring in dogs.
Similarly, “hot spot” clustering of areas with high human Lyme disease incidence has been identified – especially in southern England (Tulloch et al, 2020). It is unknown whether similar hot spots exist for dogs and it may be that these areas are useful sentinels for geographic areas where cats and dogs are at greater risk of exposure.
In comparison to Lyme, tick-borne encephalitis cases in the UK remain uncommon, but are also rising. Between 2015 and 2023, 21 tick-borne encephalitis cases with a clinical disease were diagnosed in the UK, with 12 of these being diagnosed between 2022 and 2023. Three cases acquired the infection in the UK (Callaby et al, 2025). This is a small number, but demonstrates the importance of ongoing surveillance programmes – especially as up to 60% of encephalitis cases in the UK have unknown causes, so this may well be an underestimate (Kennedy et al, 2017). Some small Babesia species are zoonotic, such as Babesia microti and Babesia venatorum, but these do not typically infect cats and dogs.
Dermacentor reticulatus ticks are the European vector for Babesia canis, a cause of potentially fatal haemolytic anaemia and thrombocytopenia in dogs. The distribution of B canis is closely linked with D reticulatus and both are increasing their range and prevalence across Europe (Figure 1).
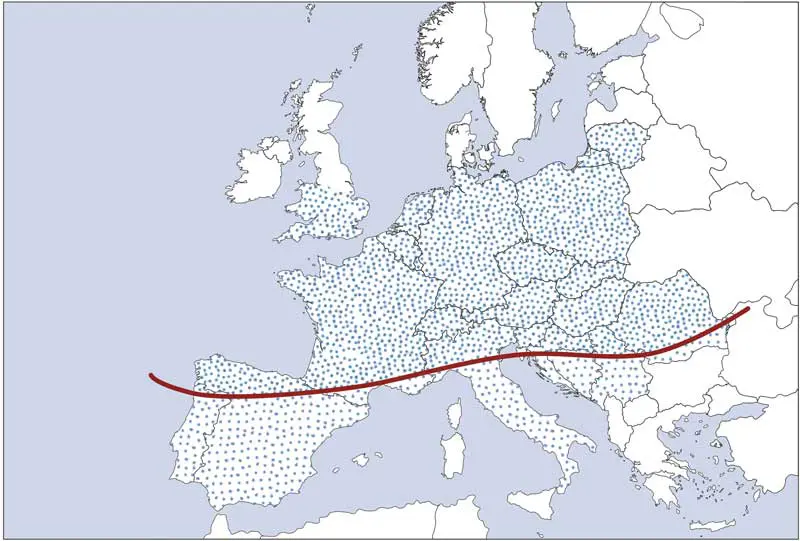
Field surveys conducted in England and Wales between 2009–2016 confirmed the presence of D reticulatus in four main areas: western Wales, north Devon, south Devon, and Essex. Unlike mainland Europe, these populations have remained mainly static, although a recent paper suggested some localised expansion in south-eastern foci (Biddlecombe et al, 2025).
This demonstrates the need for ongoing surveillance to keep track of D reticulatus populations in the UK.
Babesia canis infection can lead to life-threatening immune-mediated haemolytic anaemia and immune-mediated thrombocytopenia, with severity depending on an individual dog’s genetic susceptibility, immune status and previous exposure. Dogs also commonly present with non-specific signs such as anorexia, lethargy, weakness, pyrexia and weight loss.
The presence of D reticulatus endemic foci in England and Wales makes the UK susceptible to endemic pockets of B canis developing. This has already been demonstrated when an outbreak of babesiosis occurred in dogs from Essex between November 2015 and February 2016, when four cases of babesiosis caused by B canis were identified in untravelled dogs. Ticks recovered from infected dogs were confirmed by PCR to be D reticulatus infected with B canis (Phipps et al, 2016). To investigate this outbreak, a tick survey was carried out in the area where all four dogs had been regularly walked and D reticulatus infected with B canis was found (Hansford et al, 2016). This established the presence of an infected population of D reticulatus ticks in the proximity of the infected dogs and the likely source of infection.
A recent survey of D reticulatus ticks in the UK has found the Essex foci, but a positive tick has been found in the south-west of England (Sands et al, 2022). This demonstrates the potential for B canis to establish and spread in the UK. Vigilance for relevant clinical signs of babesiosis is, therefore, important in dogs entering the UK from abroad and those from areas endemic for D reticulatus ticks.
Rhipicephalus sanguineus is not currently endemic in the UK, as the climate is not favourable for establishment of an endemic population.
The tick is endemic in southern and eastern Europe, and moving northwards as climatic conditions become more favourable and movement of pets and wildlife reservoirs facilitates its spread (Figure 2). It has the potential to complete its life cycle rapidly, and larvae, nymphs and adults are all able to feed on pets and humans. This allows it to establish in households in a similar way to fleas.
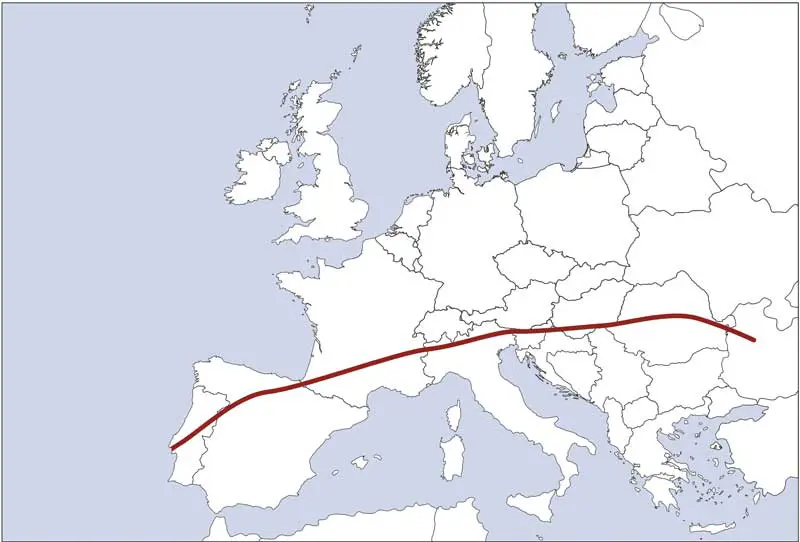
Cases of R sanguineus ticks imported on dogs from Spain, Greece and Africa with subsequent household infestations have all been reported to the European Scientific Counsel Companion Animal Parasites (ESCCAP) UK and Ireland in recent years.
In addition to the potential for house infestation, R sanguineus also carries a number of tick-borne pathogens of veterinary and/or zoonotic significance. Examples include the following.
Ehrlichia canis. Infection can lead to acute disease with pyrexia, anorexia and lymphadenopathy. A subclinical period follows where the organism is either eliminated or a chronic infection develops, with the potential for chronic disease to develop. The chronic form carries a poorer prognosis, with leukopenia, thrombocytopenia and associated immunosuppression and bleeding disorders.
Hepatozoon canis. Infections may be subclinical, but can lead to lethargy, pyrexia, anorexia, muscle wasting, multicentric lymphadenopathy, and anaemia.
Rickettsia conorii. This is zoonotic and one of several zoonotic Rickettsia species that R sanguineus can transmit. This is an issue in household infestations where humans may be bitten and disease transmitted.
Veterinary professionals and pet owners should, therefore, be vigilant for ticks on pets living or visiting central, eastern and southern Europe, and identify any found.
Changes are driven by a combination of multiple factors, including the following.
The potential severity and zoonotic nature of Lyme disease makes promoting awareness and prevention of the disease vital in dogs and owners whose lifestyle may put them at increased risk. This includes those who spend time walking in rural areas, farmland, bracken and deer-inhabited woodland.
Preventive measures are also warranted in cats whose lifestyle may put them at high risk of tick exposure, such as those with a history of tick exposure or visiting burrows and nests as part of hunting activity.
Tick prevention is essential for pets at risk of exposure. Some pets will be at considerably higher risk of exposure based on geographic location and lifestyle. A risk assessment should, therefore, be made to establish if tick prevention is required in an individual pet. Several factors should be considered.
If, after risk assessment, it is decided that tick prevention should be employed, then this should consist of a preventive product and checking for ticks after outdoor activity.
Preventive tick treatments. The use of licensed compounds that rapidly kill or repel ticks are useful in reducing tick feeding and, therefore, transmission of infection. It is important to consider compliance when discussing which product to use, as well as lifestyle. Whether a client prefers or can administer a tablet, collar or spot-on should be established, as well as whether the pet has had reactions to products in the past. Frequent swimming or bathing may make some topical products unsuitable. It should also be remembered that no product is 100% effective and owners should, therefore, still be advised to check their pet for ticks after outdoor activity if possible.
Checking for ticks after every outdoor activity. In fractious patients, TSS data suggests that the majority of ticks are located and found around the head, face, legs and ventrum (Wright et al, 2018). Nymphs are extremely small and are easily missed, so tick removal alone should not be relied upon. Ticks should be removed with a tick removal device or fine-pointed tweezers. After ticks have been removed, they should be identified, either by sending them to the TSS or by using keys such as the one on the ESCCAP UK and Ireland website. Sticking to paths where possible will greatly reduce the risk of tick exposure.
Ticks and their pathogens are a growing threat to UK pets, both in terms of increased potential exposure to ticks and from increased pet movement.
The rapidly changing distribution of ticks across Europe and tick-borne disease is reflected in the UK. Both surveillance and identification of pets and owners at high risk of exposure, therefore, remains vital if disease rates are to be reduced and if the introduction of new pathogens into the UK is to be avoided.
Ian Wright is a practising vet and co-owner of The Mount Veterinary Practice in Fleetwood, Lancashire. He has a master’s degree in veterinary parasitology and is chairman and director of the European Scientific Counsel Companion Animal Parasites (ESCCAP), as well as guidelines director for ESCCAP Europe.