31 Mar 2026
Kit Sturgess MA, VetMB, PhD, CertVR, DSAM, CertVC, FRCVS examines diet, hydration, earlier diagnosis and management options for this common issue in older felines

Figure 1. A 16-year-old neutered male cat with non-progressive chronic kidney disease (International Renal Interest Society stage 1-2). Creatinine and urine specific gravity aged 8.5 years = 149µmol/L, 1.033; aged 16 years old = 137µmol/L, 1.020.
Chronic kidney disease (CKD) remains one of the main causes of death in older cats. It is estimated that between 1% and 4% of cats in the general population have evidence of CKD, with this rising to 30% to 40% in cats older than 10 years and as high as 80% in cats aged 15 years or older (Figure 1).
A VetCompass study (Conroy et al, 2019) estimated the prevalence of CKD in UK cats at 1.2% (approximately 125,000 cats), with two-thirds having clinical signs at the time of diagnosis and a median survival time of just more than a year (interquartile range 88 to 1,042 days). This suggests that developing some level of renal dysfunction is a normal ageing process in cats and that many senior cats and most geriatric cats require some level of management of their CKD.
CKD is not a homogeneous disease, but the end point of renal insults that have led to the loss of functioning nephrons. It is generally believed that a tipping point exists in CKD such that once more than a certain amount of functional renal mass is lost, progression is inevitable, with hyperfiltrating remaining nephrons unable to sustain their viability long term (Figure 2).
The fact that CKD is an end-point diagnosis associated with varying degrees of loss of the many functions that the kidneys have, means that a single static treatment regime is not appropriate for all cats with CKD; that is, an individualised, dynamic plan needs to be developed. Such a disease-based plan can be best developed by having as full an understanding of that cat’s CKD as possible. This is an important initial discussion with the owner or carer about how much investigation should be undertaken now and what is an appropriate recheck schedule.
For many cats in early CKD, progression is slow, so a recheck every six to 12 months is sufficient. In more advanced International Renal Interest Society (IRIS) stage 3 to 4 disease, checking every two to three months may become necessary.
Once disease-based options for treatment and management have been developed, they need to be blended with patient and owner factors – such as ease of medication, ability to change diet, costs and frequency of veterinary visits – to develop a patient-owner centric contextualised care plan. Given that these are older cats, their CKD needs to be managed alongside other comorbidities such as periodontal disease, osteoarthritis, hyperthyroidism or hypertrophic cardiomyopathy.
It is also crucial to make sure the owner understands that CKD is a dynamic and progressive disease that does not follow a straight linear pathway, and that the plan will need to change as different issues and priorities arise.
Early diagnosis is an attractive proposition in cats with CKD, as it is assumed that earlier intervention will lead to slower progression (or even the possibility of arresting progression) and increased longevity.
The stumbling blocks to early diagnosis are readily available, non-invasive low cost tests that evaluate renal function prior to increases in urea and creatinine that only occur with the loss of 70% to 75% of renal mass.
The IRIS guidelines define stage 1 CKD as cats with a creatinine of less than 140µmol/L (within reference interval) and a SDMA of less than 18µg/dL (within or mildly above reference interval), with some other renal abnormality present, such as inadequate urinary concentrating ability without identifiable non-renal cause, abnormal renal palpation or renal imaging findings, proteinuria of renal origin or abnormal renal biopsy results.
This definition illustrates the lack of a reliable, straightforward, inexpensive test that defines stage 1 CKD cases, emphasising the challenge of diagnosis.
Unless some other factor is present, such as genetic or familial predisposition, it is hard to justify putting a cat showing no clinical signs through significant investigation on the off chance that stage 1 disease is present.
Apart from “big kidney, little kidney” cases or polycystic kidney disease, most cats with stage 1 disease do not have palpable or clear imaging changes. For the vast majority of cats with stage 1 disease, urine concentrating ability is retained and renal protein loss lies within the reference interval. Most cats are, therefore, diagnosed with stage 1 disease by happenstance in the course of an investigation of another issue or follow up on mild changes on routine health screening or preoperative blood tests.
This challenge also exists for cats in early stage 2 disease where, for example, a blood test has highlighted a creatinine of more than 140µmol/L or SDMA of more than 18µg/dL. Like all biochemical tests, creatinine and SDMA show interassay variability by as much as 10% to 15%, so a creatinine of 148µmol/L could give a result of 125µmol/L on a subsequent test without a change in renal function. Both parameters can also be affected by pre-renal factors such as dehydration or hypovolaemia.
The value of dietary change in early CKD remains controversial (Polzin and Churchill, 2016; Scherk and Laflamme, 2016), but some studies appear to support change even at an early stage (Coyne et al, 2026).
The optimal diet in early disease is unknown, with some evidence supporting the benefits of higher protein diets aimed at limiting sarcopenia, though others suggest that benefits exist in a more “traditional” low protein, low phosphate diet.
The reality is that a single diet optimal for all stages of CKD, or all cats even within a stage of CKD, is unlikely to exist because of their varying pathogenesis. This has led some manufacturers to develop early and later-stage CKD diets. When considering whether dietary therapy is appropriate for an individual case, the key considerations are to find a diet that the cat will eat consistently, maintaining an appropriate calorie and micronutrient intake, resulting in a stable bodyweight and minimising sarcopenia.
Following a diagnosis of early CKD, the aim is to preserve remaining glomeruli and prevent acute on chronic injury. This means:
Clear evidence exists that dietary modification improves longevity in cats with IRIS stage 3 and 4 disease, and strong support for the benefit of dietary change in cats with later IRIS stage 2 disease. Most commercial renal diets are based on protein restriction primarily as a means to manage phosphate content of the diet, although lower protein also reduces the level of uraemia.
Potassium levels are relatively increased to ensure adequate intake, as potassium-deficient acidifying diets have been shown to induce kidney damage, even in normal cats. Whatever diet is chosen, it is critical that the cat consumes adequate calories to ensure appropriate levels of micronutrients, maintenance of bodyweight and minimisation of sarcopenia. Minimising sarcopenia is important, as muscle mass has an important role in non-renal management of nitrogenous waste.
Other features of a renal diet include:
A wide variety of nutraceuticals and dietary supplements have been suggested and undergone limited clinical trials. To date, evidence supporting their use is poor, with trials focusing on short-term measures rather than long-term impact such as longevity. However some have at least reasonable therapeutic rationale and could be considered once a management strategy based on most urgent need has been developed.
Maintaining hydration is not usually a significant issue in stage 1 and 2 CKD, but as renal concentrating ability fails, although the patient may be polydipsic, the polyuria may exceed this.
Estimating fluid requirement can be tricky, as standard values may not apply. Maximising intake in food is the first step, which is why wet food is a good option for CKD cats – especially those that do not seem to like to drink. Ensuring easy access to water of a type the cat likes will also help, whether that be a dripping tap or toilet.
The value of water fountains is unclear and has not been shown to increase water intake in normal cats (Grant, 2010). For some cats, providing gravy or liquid feed may help, but for those cats not maintaining hydration orally then subcutaneous fluid can be considered.
It is difficult to judge how much fluid is needed, but a rough calculation can be made on water consumption plus moisture in food consumed matched against daily requirement, which is likely to be around 70ml/kg to 100ml/kg for a cat with stage 3 disease. This can mean the patient needs 100ml to 200ml of additional fluid daily. No studies have been conducted looking at optimal fluid composition, but both 0.9% saline and Hartmann’s (0.6% sodium chloride) can lead to sodium overload, so for long-term use, 0.45% saline with 2.5% glucose is a reasonable choice. For many cats, once a subcutaneous pocket has been created for fluid administration, repeating the fluid only causes minor discomfort.
Potential drug therapies for managing CKD in cats are many, and it is easy to overwhelm an owner and the patient with the amount of medication they are expected to give.
Beyond the caregiver burden, the risk is that the owner will select the medications they find easiest to give, which may not be the ones that are most beneficial for the patient. If multiple medications are prescribed, it is often worth ranking them for the owner and making sure they understand that it is okay to contact the team for help and support if they are struggling. When ranking therapies, start with those where the most evidence of ability to increase longevity and/or improved quality of life is available.
The key factors that may need to be addressed in cats with CKD are:
Weight loss and a lack of adequate nutritional intake is the factor that most commonly precipitates a decision to euthanise a CKD cat that is not in crisis.
Reduced intake is likely to be multifactorial, associated with dietary palatability, nausea and hyporexia. Caloric intake can be improved by appetite stimulation and management of nausea. In the UK, mirtazapine is the best appetite stimulant available, but elsewhere, a ghrelin agonist (capromorelin; Wofford et al, 2025) is available which for many cats may be more effective, with fewer side effects.
Control of nausea can require a number of different medications to be tried; anecdotally, the most effective seem to be maropitant and ondansetron for chronic use.
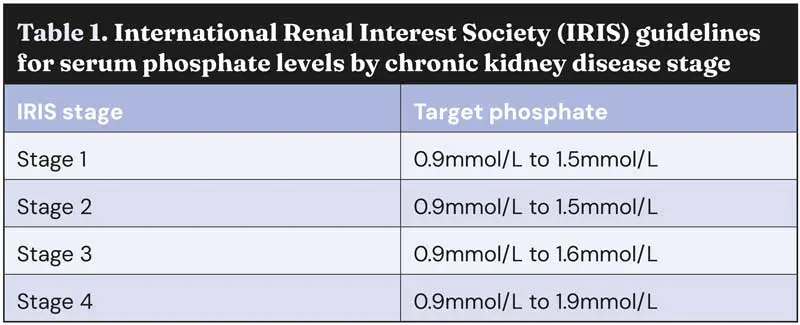
As elevated phosphate has been shown to reduce life expectancy, trying to manage phosphate to within IRIS guidelines is a key goal.
In early stages, phosphate management may not be required, even in cats on a standard diet. However, both PTH and FGF-23 are elevated in advance of changes in phosphate (Lin et al, 2021) being outside the target range and FGF-23 levels correlate with survival. As yet, it has not been demonstrated that reducing FGF-23 by phosphate restriction when serum phosphate is within target range leads to improved survival. Elevated FGF-23 may, however, be a useful indicator that managing phosphate would be beneficial and prompt dietary intervention earlier in CKD cases.
If dietary management does not lead to phosphate control, phosphate binders should be considered (Table 2). A variety of options are available but can be challenging to use due to tablet size, frequency of dosing and palatability.
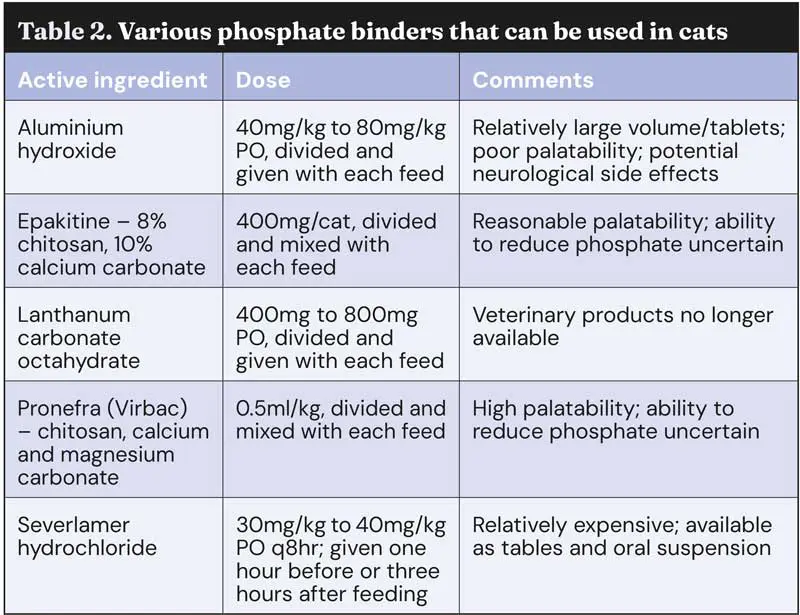
Unfortunately, some of the more palatable options have significantly less impact on serum phosphate levels. It is important that giving phosphate binders are associated with feeding, as they act to limit phosphate absorption by the gastrointestinal tract; this makes their use tricky in cats fed ad lib.
Potassium intake requirement is not an absolute and will depend on the level of protein in the diet, whether the diet is acidifying or not, and the level of polyuria/polydipsia and acid-base status of the patient.
The frustration when trying to determine whether potassium supplementation is required is that assessing whole-body potassium is not practical, as most potassium is intracellular. This is further complicated by acid-base status, as acidosis tends to promote intracellular potassium being swapped for hydrogen ions, which can make serum potassium appear adequate despite a significant whole-body deficit.
Failure to meet a patient’s potassium requirements can lead to more rapid progression. Where possible, acid-base status should be assessed for patients with acidosis and hypokalaemia; potassium citrate can be used to manage both issues, whereas if it is only potassium that is needed, potassium gluconate is an alternative. In cats, potassium chloride supplements should be avoided.
Where potassium supplementation beyond diet is required, this will be 2mmol to 4mmol (mEq) of potassium/day for most cats. Lots of supplements are available online, but many contain inadequate potassium levels to achieve this dose, so it is critical to check the label if the client is sourcing their own potassium supplements.
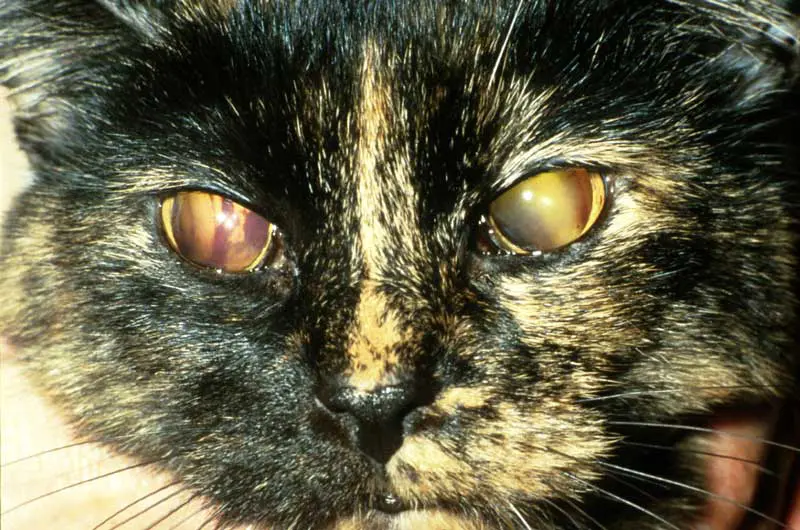
The presence of significant proteinuria is associated with more rapid progression of CKD. Hypertension impacts quality of life and risks serious adverse events such as retinal detachment, hyphaema and blindness (Figure 3), although managing hypertension, to date, has not resulted in increased longevity.
In terms of treatment, the two most commonly used anti-hypertensive drugs are calcium channel blockers (amlodipine) or angiotensin receptor blockers (ARB; telmisartan); for proteinuria, angiotensin-converting enzyme inhibitors or ARB.
Recommendations for first choice will depend on availability, cost and ease of medication, as no published studies directly compare their efficacy. However, if the patient is both hypertensive and proteinuric, then using telmisartan as a single medication in the first instance reduces cost and medication requirements.
Many mechanisms exist that can lead to anaemia in cats with CKD, one of which is a lack of bone marrow stimulation and production. No precise figure exists at which management of anaemia associated with lack of red cell production is required. Generally, this should be when evidence exists that the anaemia of itself is affecting quality of life. This usually means that the anaemia is at least moderate (packed cell volume [PCV] or haematocrit less than 18%), but assessing whether a lack of appetite, lethargy, or inability to jump on to surfaces is the result of anaemia or a different facet of the patient’s CKD is challenging. So, for those cats in which red cell counts are at a level where clinical signs are likely, a treatment trial is reasonable to see whether the signs resolve as the PCV/haematocrit rises or not.
Options vary with drug availability; traditionally, this has been with erythropoietin or erythropoietin analogues (darbepoetin), but more recently another key player in bone marrow production of red cells, hypoxia-induced factor (HIF), has become a viable target. Molidustat blocks the degradation of HIF, resulting in improved red cell production, and is available as an oral preparation for use in cats in some areas of the world, providing a good alternative to darbepoetin that can be administered by the owner at home.
Managing comorbidities such as osteoarthritis, dental disease, hypertrophic cardiomyopathy and hyperthyroidism is an important part of delivering optimal care.
Decisions on treatment recommendations need to be taken on a whole patient basis; that is, benefits and risks of the treatment on all of the known conditions. This sometimes means that a compromise needs to be made and should be based on the most critical current challenges, both medically and from an owner-patient management perspective.
It is important to make sure that the owner is aware that no perfect solution exists and treatment for one issue may impact other diseases the cat has.
Understanding as much as possible about an individual cat’s CKD is an important first step in delivering optimal treatment for the patient and owner.
Once the key challenges have been identified, this will highlight which treatments are likely to be most valuable in improving quality of life and longevity.
Early diagnosis can be challenging, but a mounting body of evidence suggests that earlier intervention will slow the rate of progression of the cat’s CKD, which will also lead to an improved quality of life and longevity.
Use of some of drugs is under the veterinary medicine cascade.
Kit Sturgess graduated from the University of Cambridge in 1986, then spent six years in general veterinary practice. He has further professional qualifications in imaging, cardiology and internal medicine, as well as a PhD for looking at the effects of FIV on mucosal immune function. Kit is a fellow of the RCVS, a specialist in small animal medicine and an advanced practitioner in veterinary cardiology. He has been seeing referral small animal medicine cases for the past 25 years – both at university-based and private specialist practices. Kit’s love of teaching and learning led him to develop a new, more flexible, role combining lecturing, writing and clinic time. The majority of his clinical time is spent providing an internal medicine service at Optivet Referrals in Havant, Hampshire. Kit maintains a keen interest in many areas of internal medicine and has authored numerous articles and two textbooks, and presented lectures and research abstracts at conferences worldwide.